
AlphaLISA SureFire Ultra Human and Mouse Total FOXO1 Detection Kit, 50,000 Assay Points
The AlphaLISA™ SureFire® Ultra™ Human and Mouse Total FOXO1 assay is a sandwich immunoassay for quantitative detection of total FOXO1 in cellular lysates using Alpha Technology.
| Feature | Specification |
|---|---|
| Application | 細胞シグナル伝達 |
| Protocol Time | 2h at RT |
| Sample Volume | 10 µL |
The AlphaLISA™ SureFire® Ultra™ Human and Mouse Total FOXO1 assay is a sandwich immunoassay for quantitative detection of total FOXO1 in cellular lysates using Alpha Technology.



Product information
Overview
Forkhead Box O1 (FOXO1) is a transcription factor that regulates genes involved in glucose metabolism, cell cycle arrest, apoptosis, and oxidative stress resistance. FOXO1 activity is controlled by AKT-mediated phosphorylation, which promotes nuclear exclusion and proteasomal degradation. Dephosphorylated FOXO1 translocates to the nucleus and activates target genes including glucose-6-phosphatase, PEPCK, and p27Kip1. FOXO1 plays critical roles in hepatic gluconeogenesis and functions as a tumor suppressor in cancer. Dysregulation of FOXO1 is implicated in type 2 diabetes and cancer, making it a therapeutic target for metabolic disorders and malignancies.
The AlphaLISA SureFire Ultra Human and Mouse Total FOXO1 is a sandwich immunoassay for the quantitative detection of total FOXO1 in cellular lysates, using Alpha Technology.
Formats:
- The HV (high volume) kit contains reagents to run 100 wells in 96-well format, using a 60 μL reaction volume.
- The 500-point kit contains enough reagents to run 500 wells in 384-well format, using a 20 μL reaction volume.
- The 10,000-point kit contains enough reagents to run 10,000 wells in 384-well format, using a 20 μL reaction volume.
- The 50,000-point kit contains enough reagents to run 50,000 wells in 384-well format, using a 20 μL reaction volume.
AlphaLISA SureFire Ultra kits are compatible with:
- Cell and tissue lysates
- Antibody modulators
- Biotherapeutic antibodies
AlphaLISA SureFire Ultra kits can be used for:
- Cellular kinase assays
- Receptor activation studies
- High-throughput screening for preclinical studies
How it works
Total-AlphaLISA SureFire Ultra assay principle
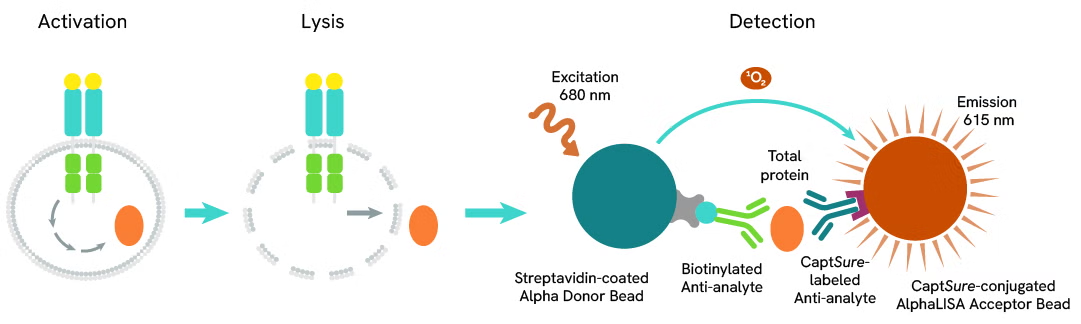
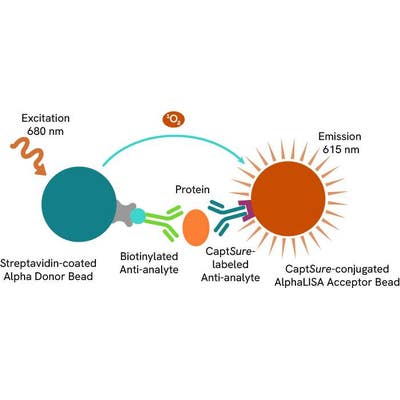
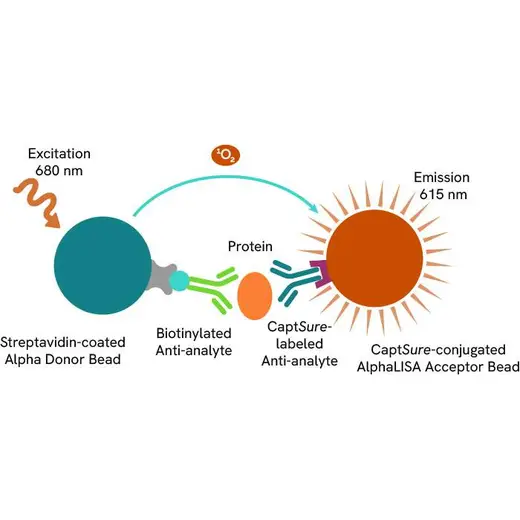
The Total-AlphaLISA SureFire Ultra assay measures the expression level of a protein target in a cell lysate.
The Total-AlphaLISA SureFire Ultra assay uses two antibodies which recognize two different distal epitopes on the targeted protein. AlphaLISA assays require two bead types: Acceptor and Donor beads. Acceptor beads are coated with a proprietary CaptSure™ agent to specifically immobilize the assay specific antibody, labeled with a CaptSure tag. Donor beads are coated with streptavidin to capture one of the detection antibodies, which is biotinylated. In the presence of targeted protein, the two antibodies bring the Donor and Acceptor beads in close proximity whereby the singlet oxygen transfers energy to excite the Acceptor bead, allowing the generation of a luminescent Alpha signal. The amount of light emission is directly proportional to the quantity of protein present in the sample.

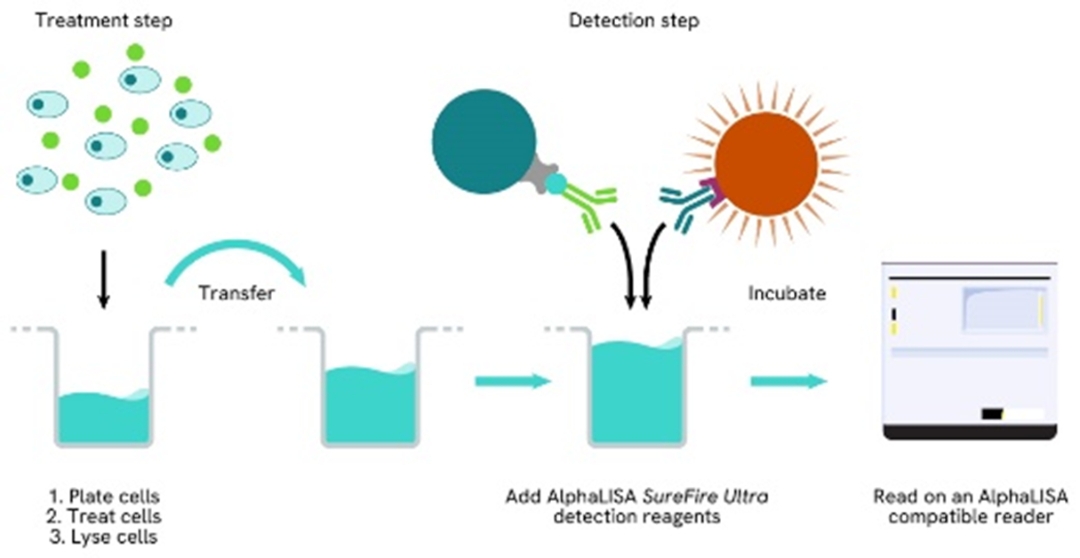
Total-AlphaLISA SureFire Ultra two-plate assay protocol
The two-plate protocol involves culturing and treating the cells in a 96-well plate before lysis, then transferring lysates into a 384-well OptiPlate™ plate before the addition of Total-AlphaLISA SureFire Ultra detection reagents. This protocol permits the cells viability and confluence to be monitored. In addition, lysates from a single well can be used to measure multiple targets.
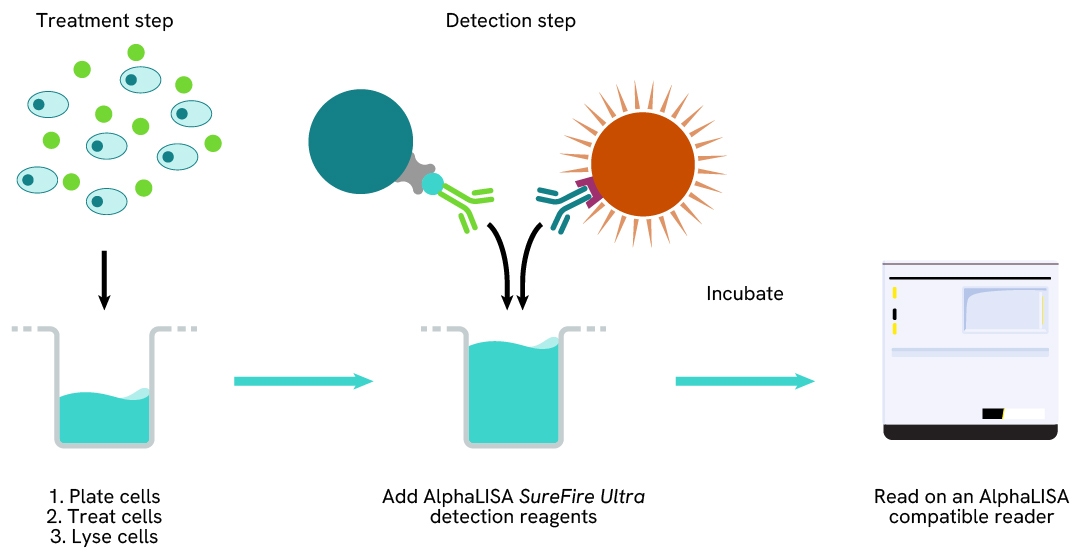
Total-AlphaLISA SureFire Ultra one-plate assay protocol
Detection of Total target protein with AlphaLISA SureFire Ultra reagents can be performed in a single plate used for culturing, treatment, and lysis. No washing steps are required. This HTS designed protocol allows for miniaturization while maintaining AlphaLISA SureFire Ultra quality.
Assay validation
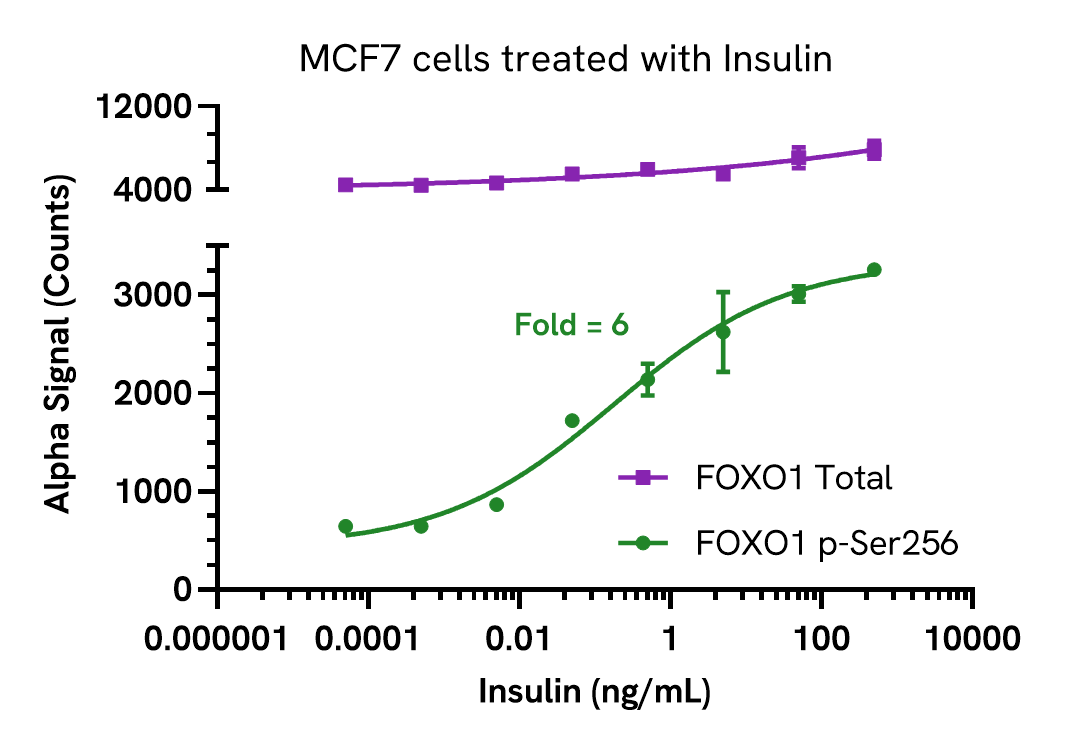
Validation of FOXO1 Total in cells treated with insulin
MCF7 cells were seeded in a 96-well plate (20,000 cells/well) in complete medium, and incubated overnight at 37°C, 5% CO2. The cells were starved for 2 hours and treated with increasing concentrations of insulin for 5 minutes prepared in serum free medium.
After treatment, the cells were lysed with 100 µL of Lysis Buffer for 10 minutes at RT with shaking (350 rpm). FOXO1 Phospho (Ser256) and Total levels were evaluated using respective AlphaLISA SureFire Ultra assays. For the detection step, 10 µL of cell lysate (approximately 4,000 cells) was transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor mix and incubated for 1 hour at RT. Finally, 5 µL of Donor mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
As expected, insulin triggered a dose-dependent increase in the levels of Phospho FOXO1 (Ser256) with no significant change in Total levels.
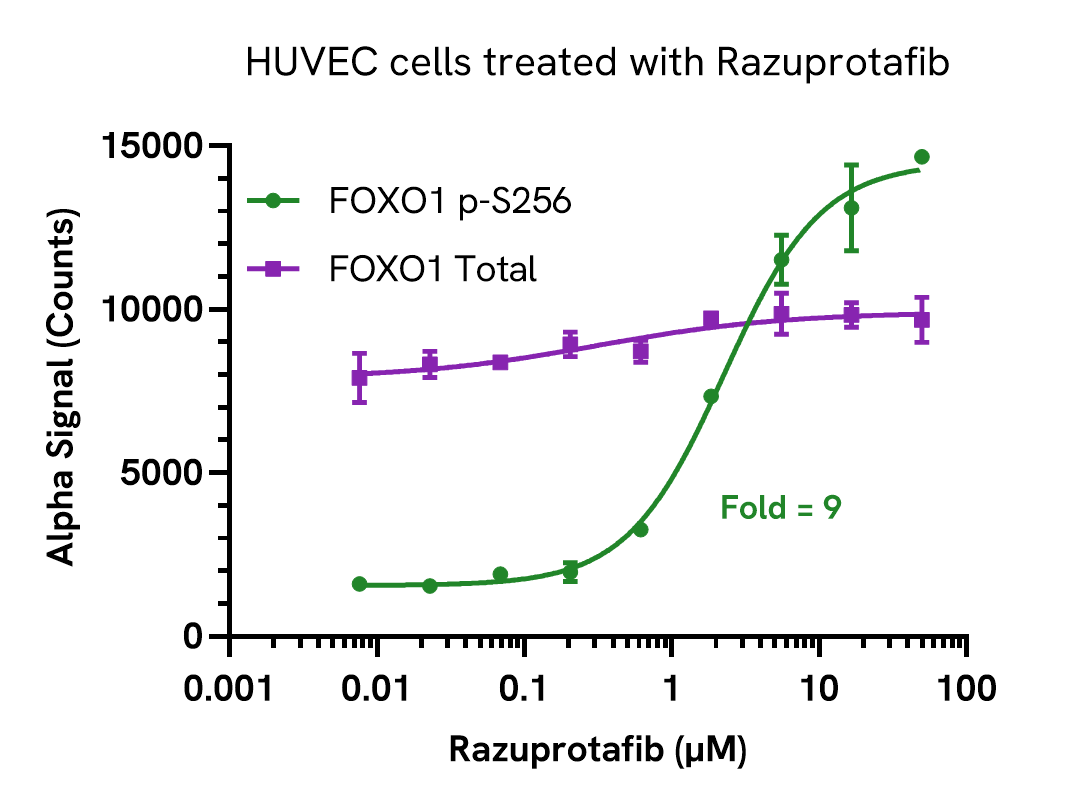
Validation of FOXO1 Total in HUVEC cells treated with Razuprotafib
HUVEC cells were seeded in a 96-well plate (20,000 cells/well) in complete medium, and incubated overnight at 37°C, 5% CO2. The cells were starved for 2 hours and treated with increasing concentrations of Razuprotafib for 15 minutes.
After treatment, the cells were lysed with 25 µL Lysis Buffer for 10 minutes at RT with shaking (350 rpm). FOXO1 Phospho (Ser256) and Total levels were evaluated using respective AlphaLISA SureFire Ultra assays. For the detection step, 10 µL of cell lysate (approximately 12,000 cells) was transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor mix and incubated for 1 hour at RT. Finally, 5 µL of Donor mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
Razuprotafib treatment results in the activation of the Tie2 signaling pathway in endothelial cells and a dose-dependent increase in the levels of Phospho FOXO1 (Ser256) with no significant changes in Total levels.
Assay specificity/selectivity
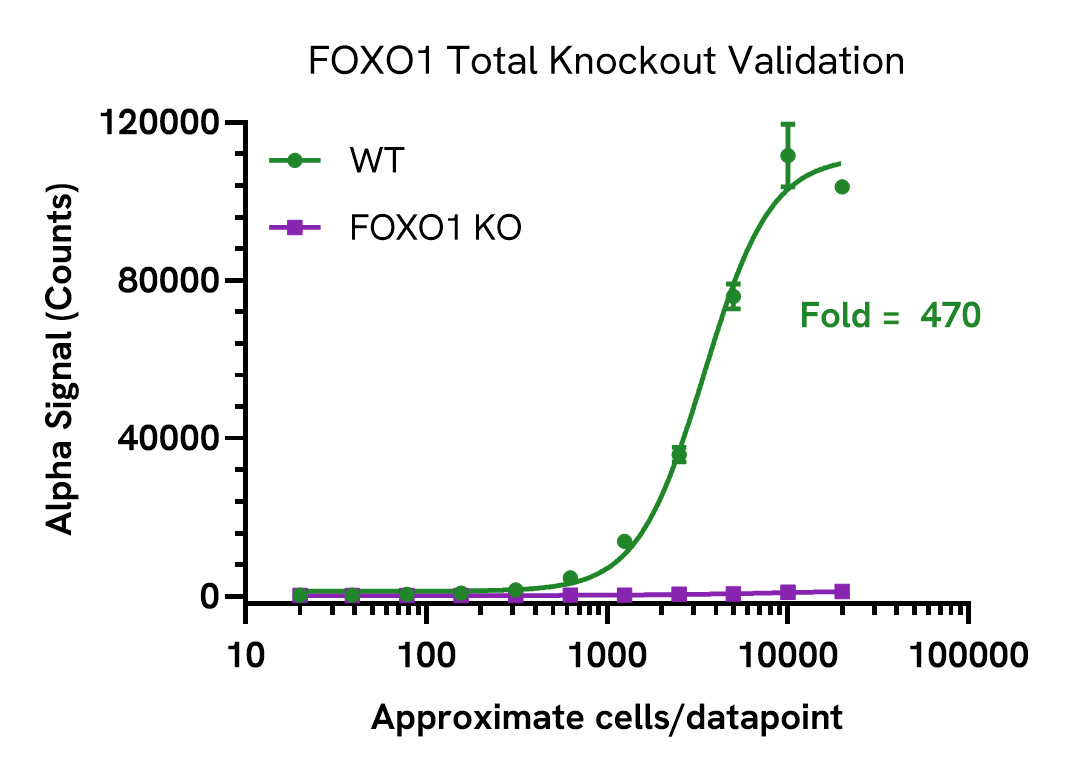
Knockout validation of FOXO1 Total assay
Total FOXO1 levels were assessed in HAP1 wild type (WT) and FOXO1 knockout (KO) cells. Cells were cultured to confluency in T175 flasks at 37°C, 5% CO2. Cells were lysed in 4 mL of Lysis Buffer.
Lysates were serially diluted in Lysis Buffer and FOXO1 Total levels were evaluated using the AlphaLISA SureFire Ultra kit.
For the detection step, 10 µL of lysate was transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor mix and incubated for 1 hour at room temperature. Finally, 5 µL of Donor mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
Total FOXO1 was only detected in WT cells, confirming assay specificity.
Assay versatility
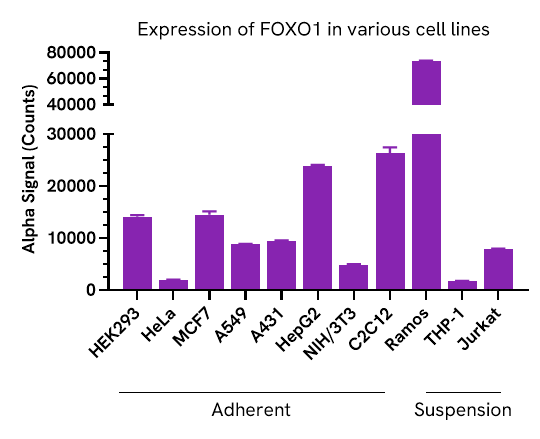
Expression of FOXO1 in various cell lines
Adherent cells were grown to confluency in a T175 flask at 37°C, 5% CO2, and were lysed with Lysis Buffer at a density of 0.5 x 106 cells/mL. Suspension cells were harvested, washed in HBSS and lysed with Lysis Buffer at 1.6 x 106 cells/mL.
FOXO1 levels were evaluated using the AlphaLISA SureFire Ultra assay. For the detection step, 10 µL of cell lysate were transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor Mix and incubated for 1 hour at RT. Finally, 5 µL of Donor Mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
Total FOXO1 protein is expressed in a wide range of cell types with high expression in Ramos, C2C12 and Hep G2 cell lines.
Assay sensitivity
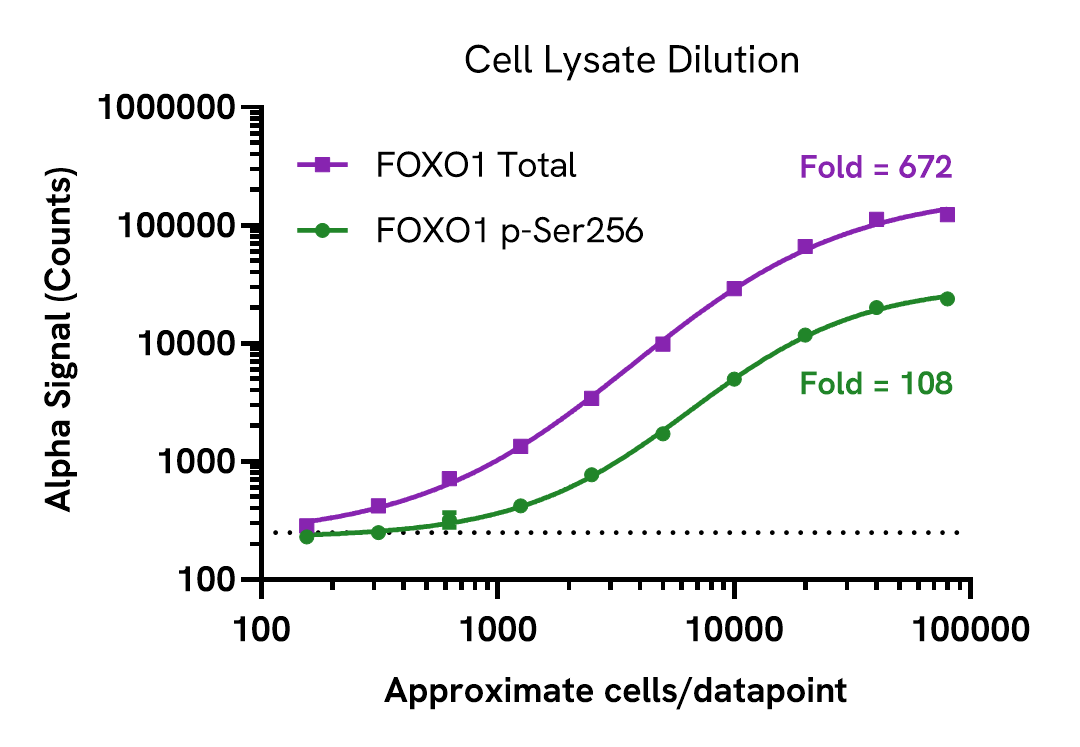
Assay sensitivity - cell lysate
Cell lysate was prepared from HEK293 cells seeded in T175 flasks and cultured to confluence and then lysed with 3 mL of Lysis Buffer for 10 minutes at RT with shaking.
Lysates were serially diluted in Lysis Buffer and assayed for Phospho (Ser256) and Total FOXO1 levels using respective AlphaLISA SureFire Ultra kits. For the detection step, 10 µL of lysate was transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor mix and incubated for 1 hour at room temperature. Finally, 5 µL of Donor mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
Approximate number of cells/datapoint is indicated on the graph. The dotted line represents assay background. This assay can detect FOXO1 expression in less than 1,250 cells/datapoint.
Specifications
| Application |
Cell Signaling
|
|---|---|
| Automation Compatible |
Yes
|
| Brand |
AlphaLISA SureFire Ultra
|
| Detection Modality |
Alpha
|
| Product Group |
Kit
|
| Protocol Time |
2h at RT
|
| Sample Volume |
10 µL
|
| Shipping Conditions |
Shipped in Blue Ice
|
| Target |
FOXO1
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
Mouse
|
| Technology |
Alpha
|
| Therapeutic Area |
Metabolic
Neuroscience
Oncology
|
| Unit Size |
50,000 Assay Points
|
Resources
Are you looking for resources, click on the resource type to explore further.
The definitive guide for setting up a successful AlphaLISA SureFire Ultra assay
Several biological processes are regulated by...


How can we help you?
We are here to answer your questions.






























