
IVISense Toxicology Fluorescent Imaging Panel
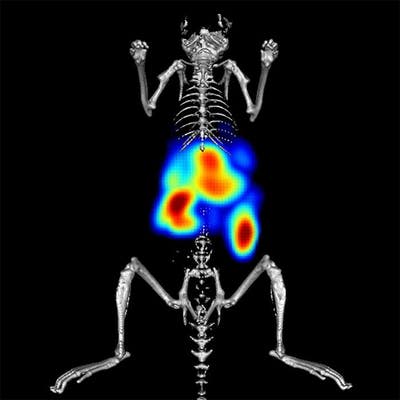
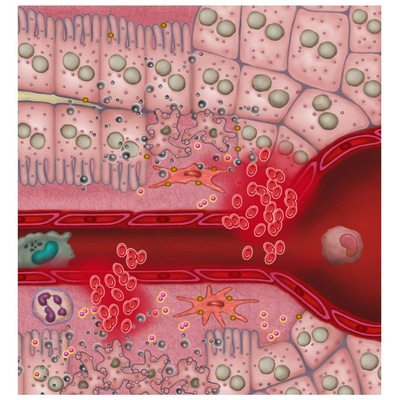
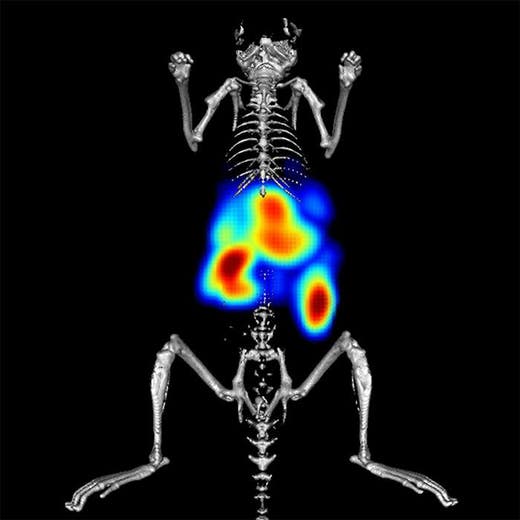
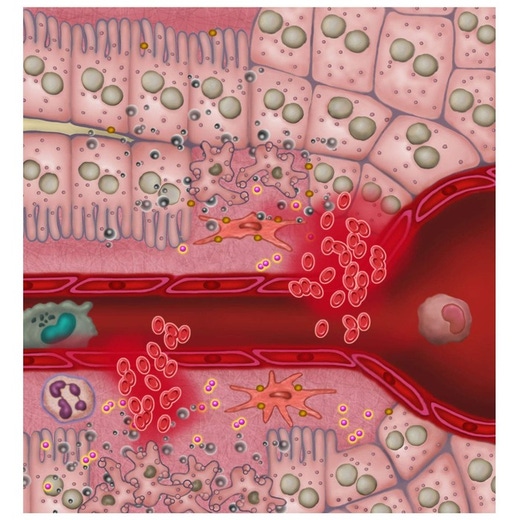


IVISense™ Toxicology Fluorescent Imaging Panel is designed to detect drug-induced liver injury (DILI) early in the drug discovery process. DILI is a leading cause of project termination. The panel allows researchers to image inflammation, tissue destruction, metabolic changes, and vascular leak (edema). By measuring these biological changes prior to overt histological signs, the panel provides predictive data on potential drug toxicity.
IVISense™ Toxicology Fluorescent Imaging Panel is designed to detect drug-induced liver injury (DILI) early in the drug discovery process. DILI is a leading cause of project termination. The panel allows researchers to image inflammation, tissue destruction, metabolic changes, and vascular leak (edema). By measuring these biological changes prior to overt histological signs, the panel provides predictive data on potential drug toxicity.








Loading...
Product information
Overview
Our Toxicology Fluorescent Imaging Panel of probes allows you to image inflammation, tissue destruction, metabolic changes, and vascular leak (edema), in a manner that can be predictive of potential drug toxicity, measuring biological changes prior to overt histological signs. Our published research shows the utility of a three-probe cocktail for imaging drug-induced liver injury (Vasquez & Peterson, J Pharmacol Exp Ther 2017; 361:87-98) via a ratio of probes specifically designed to optimize liver injury signal while minimizing normal background signal in liver and kidneys. The Toxicology Panel provides an extra vial of IVISense MMP 750 FAST to allow the preparation of the cocktail.
The inclusion of probes at 680 nm and 750 nm wavelengths further offers the opportunity for multiplex imaging of appropriate probe combinations to maximize information gained from research animals.
Additional product information
Our IVISense toxicology fluorescent imaging panel includes the following five probes (6 vials)
| Part number | Fluorescent probe | Biological target |
|---|---|---|
| NEV10054EX | IVISense Vascular 680 (AngioSense 680EX) | Vascular probe: Leaks into sites of vascular damage or inflammation |
| NEV10091 | IVISense Transferrin Receptor 750 (Transferrin-Vivo 750) | Transferrin receptor targeted probe: Iron metabolism marker in liver |
| NEV10168 x 2 | IVISense MMP 750 FAST (MMPSense 750 FAST) | Pan-MMP activatable probe: Secreted marker of stellate cells, Kupffer cells, macrophages, and neutrophils |
| NEV11053 | IVISense Annexin-V 750 Fluorescent Probe (Annexin-Vivo) | Death/apoptosis probe: Marker on inflammatory cells; metabolic marker on tumor cells |
| NEV11079 | IVISense Renin 680 FAST (ReninSense 680 FAST) | Renin activatable probe: Marker of tissue renin-angiotensin system activity |
Specifications
| Brand |
IVISense
|
|---|---|
| Imaging Modality |
Fluorescence
|
| Shipping Conditions |
Shipped in Blue Ice
|
| Storage Conditions |
+2 to +8 °C
|
| Therapeutic Area |
Toxicology
|
| Unit Size |
1 kit containing 5 vials
|
Resources
Are you looking for resources, click on the resource type to explore further.
Loading...


How can we help you?
We are here to answer your questions.






























