

HTRF Human CCL17 Detection Kit, 10,000 Assay Points

The HTRF human CCL17 kit is for the simple and rapid quantification of soluble human CCL17 proteins in cell culture supernatants, and can be used as a no-wash alternative to more traditional wash-based ELISA.
| Feature | Specification |
|---|---|
| Application | タンパク質定量 |
| Dynamic Range | 21 - 5500 pg/mL |
| Limit of Detection | 20 pg/mL |
| Limit of Quantification | 35.3 pg/mL |
| Sample Volume | 16 µL |
The HTRF human CCL17 kit is for the simple and rapid quantification of soluble human CCL17 proteins in cell culture supernatants, and can be used as a no-wash alternative to more traditional wash-based ELISA.



Product information
Overview
Human CCL17, also known as thymus and activation regulated chemokine (TARC), was initially isolated from phytohemagglutinin-stimulated peripheral blood mononuclear cells. CCL17 is constitutively expressed in the thymus and, under activation, in several cell types. CCL17-mediated recruitment of Th2 cells and CLA+ CD4+ T cells plays a key role in allergic diseases such as atopic dermatitis, allergic asthma, allergic rhinitis, and allergic contact dermatitis. In addition, CCL17 has been detected in idiopathic pulmonary fibrosis. CCL17 and CCL22 secreted by dendritic cells (DCs) seem to mediate the recruitment of regulatory T cells to sites of inflammation in patients with chronic hepatitis.
HTRF assays offer many advantages over other technologies:
- Homogeneous add-and-read format
- No wash steps
- Low background
- Straightforward miniaturization from 96- or 384-well microplates to high density assay formats such as 384-well low volume and 1536-well plates
- Stable signal, providing flexibility in time of readout or size of assays
How it works
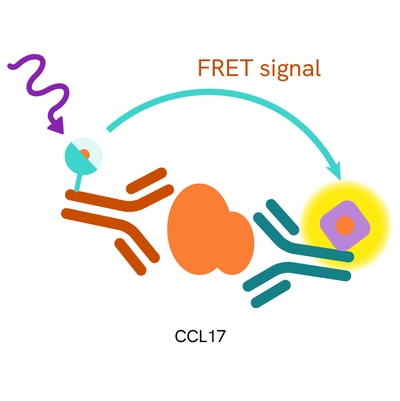
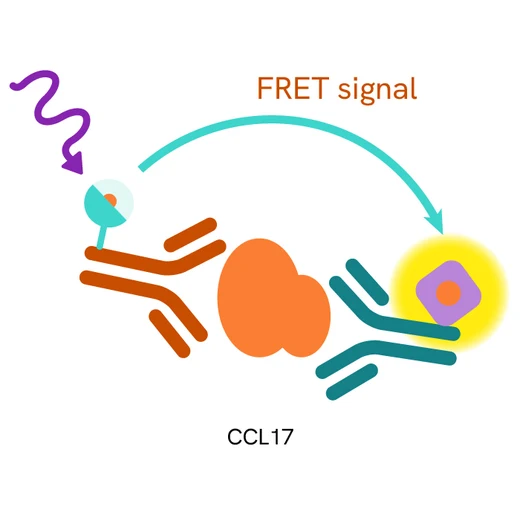
Principle of the HTRF human CCL17 assay
The HTRF human CCL17 assay is based on a TR-FRET sandwich immunoassay involving two specific antibodies, one labelled with Eu3+-cryptate (donor) and the other with d2 (acceptor). Both antibodies are directed against the CCL17 protein. They bind to soluble CCL17, and the donor-acceptor proximity leads to a fluorescent TR-FRET signal. The intensity of the signal is directly proportional to the concentration of soluble CCL17 present in the sample (cell supernatant).

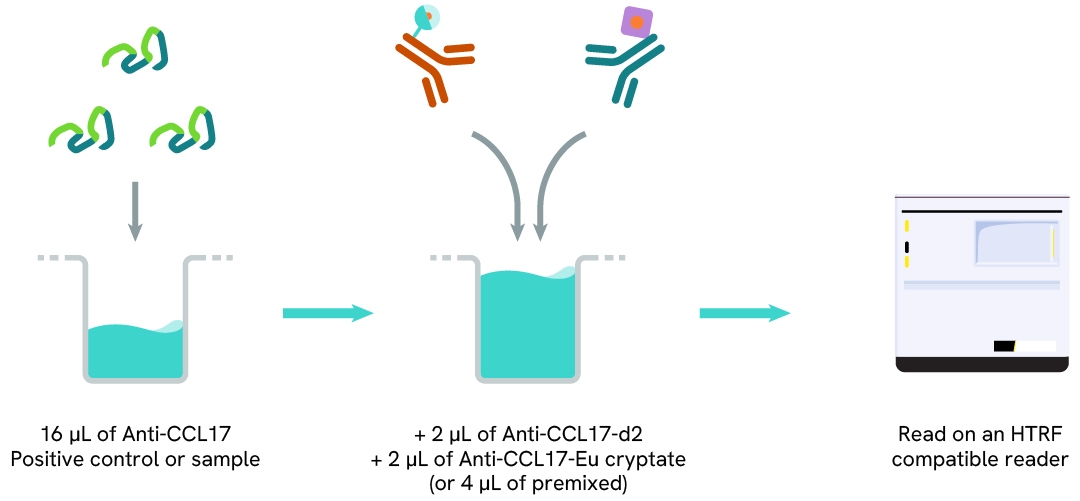
Protocol of the HTRF human CCL17 assay
The HTRF human CCL17 assay can be run in 96- or 384-well low volume white detection plates (20 µL final). As described here, samples (cell supernatants) or standards are dispensed directly into the assay plate for the detection of human CCL17 by HTRF reagents. The antibodies labelled with HTRF fluorophores may be pre-mixed and added in a single dispensing step. No washing steps are needed. The protocol can be further miniaturized or upscaled by simply resizing each addition volume proportionally.
Assay details
Human CCL17 assay details
| Sample size | 16 µL |
|---|---|
| Final assay volume | 20 µL |
| Time to result | 2h at RT |
| Kit component | Frozen detection antibodies, lyophilized standard & buffers |
| Species | Human, some cross-reactivity with mouse protein |
Analytical performance
Intra-assay precision table
Each of the 3 samples was measured 24 times, and the % CV was calculated for each sample. Samples were supernatants from PHA-treated PBMC cells.
| Sample | [CCL17] (pg/mL) | CV |
|---|---|---|
| 1 | 1874 | 8.1% |
| 2 | 924 | 7.5% |
| 3 | 501 | 10.5% |
| Mean CV | 8.7% |
Inter-assay precision table
Each of the samples was measured in 3 independent experiments (3 days), and the % CV was calculated for each sample. Samples were supernatants from PHA-treated PBMC cells.
| Sample | [CCL17] (pg/mL) | CV |
|---|---|---|
| 1 | 2,773 | 8.7% |
| 2 | 1,231 | 10.8% |
| 3 | 647 | 13.3% |
| Mean CV | 11.0% |
Spike and recovery
Each of the samples was measured in 3 independent experiments (3 days), and the % CV was calculated for each sample. Samples were supernatants from PHA-treated PBMC cells spiked with CCL17 recombinant protein.
| Spiked | % Recovery | |
|---|---|---|
| IL6 (pg/mL) | Diluent 5 | RPMI+10% FBS |
| 375 | 103% | 99% |
| 250 | 102% | 106% |
| 125 | 94% | 85% |
Dilutional linearity
Each of the samples was measured in 3 independent experiments (3 days), and the % CV was calculated for each sample. Samples were supernatants from PHA-treated PBMC cells.
| Sample dilution factor (x) | Expected [CCL17] | Observed [CCL17] | Dilution Recovery (%) |
|---|---|---|---|
| (pg/mL) | (pg/mL) | ||
| neat | 4217.3 | 3840.4 | 91% |
| 1/2 | 1692.3 | 1920.2 | 113% |
| 1/4 | 813.0 | 960.1 | 118% |
| 1/8 | 426.7 | 480.1 | 112% |
| 1/16 | 236.8 | 240.0 | 101% |
| 1/32 | 135.8 | 120.0 | 89% |
Assay validation
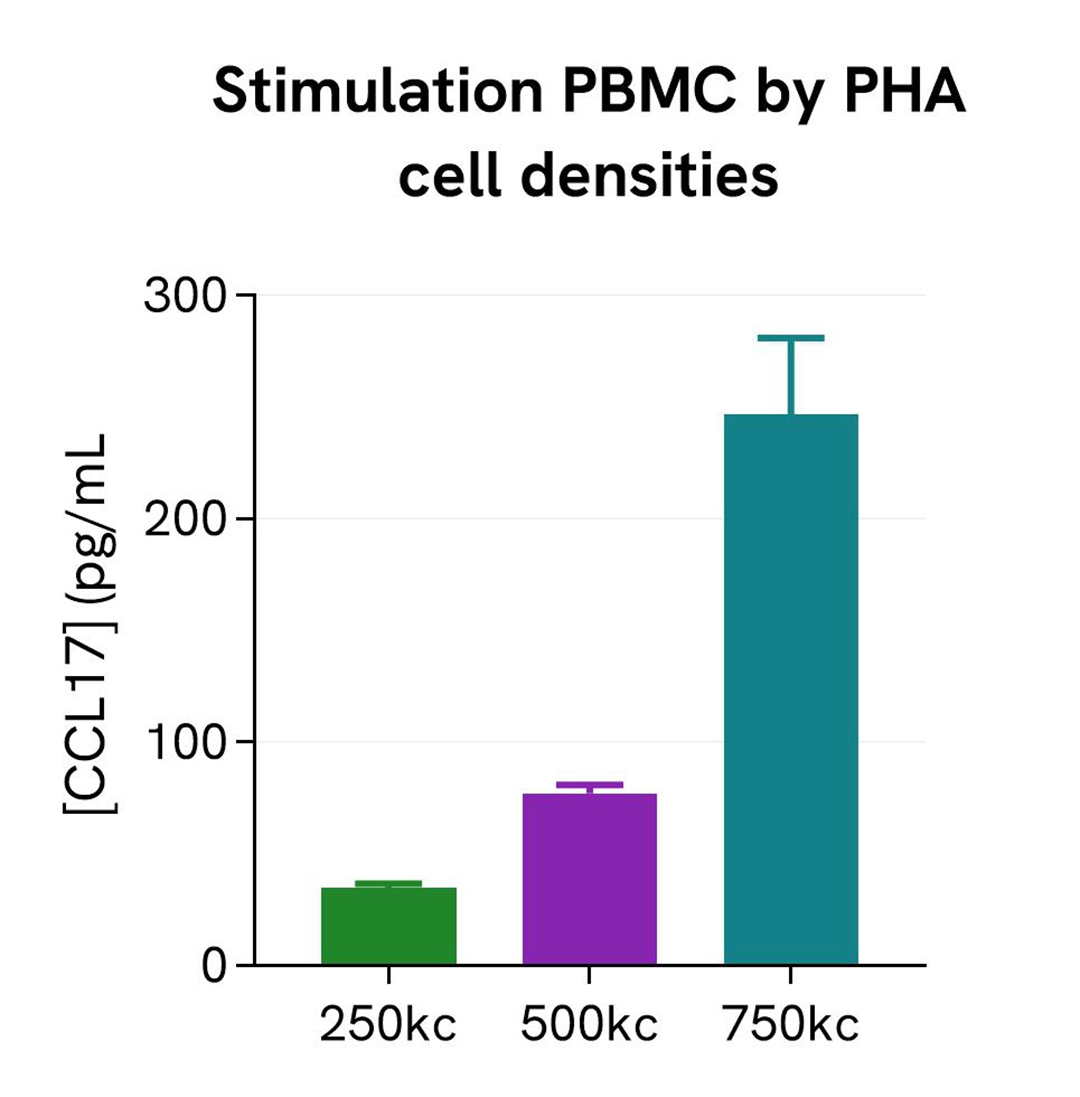
Cell densities of PHA-stimulated PBMCs
Human PBMCs were stimulated with 10 µg/mL of PHA in RPMI for 24h, in a humidified atmosphere at 37°C and 5% CO2. Different cell densities were tested: 250, 500, and 750 kcells/cell were plated in a 96-well plate.
After 24h of stimulation, the cell supernatants were collected. 16 µL of each sample were transferred into a white detection plate (384 wells, low volume) to measure the secreted CCL17 in each sample. Then 4 µL of the HTRF human CCL17 detection reagents were added to the wells. After 2h, the signals were recorded for every sample.
The CCL17 concentration in each supernatant sample was extrapolated from the standard curve prepared in RPMI.
As expected, the secretion of CCL17 increased with increasing PBMC cell density.
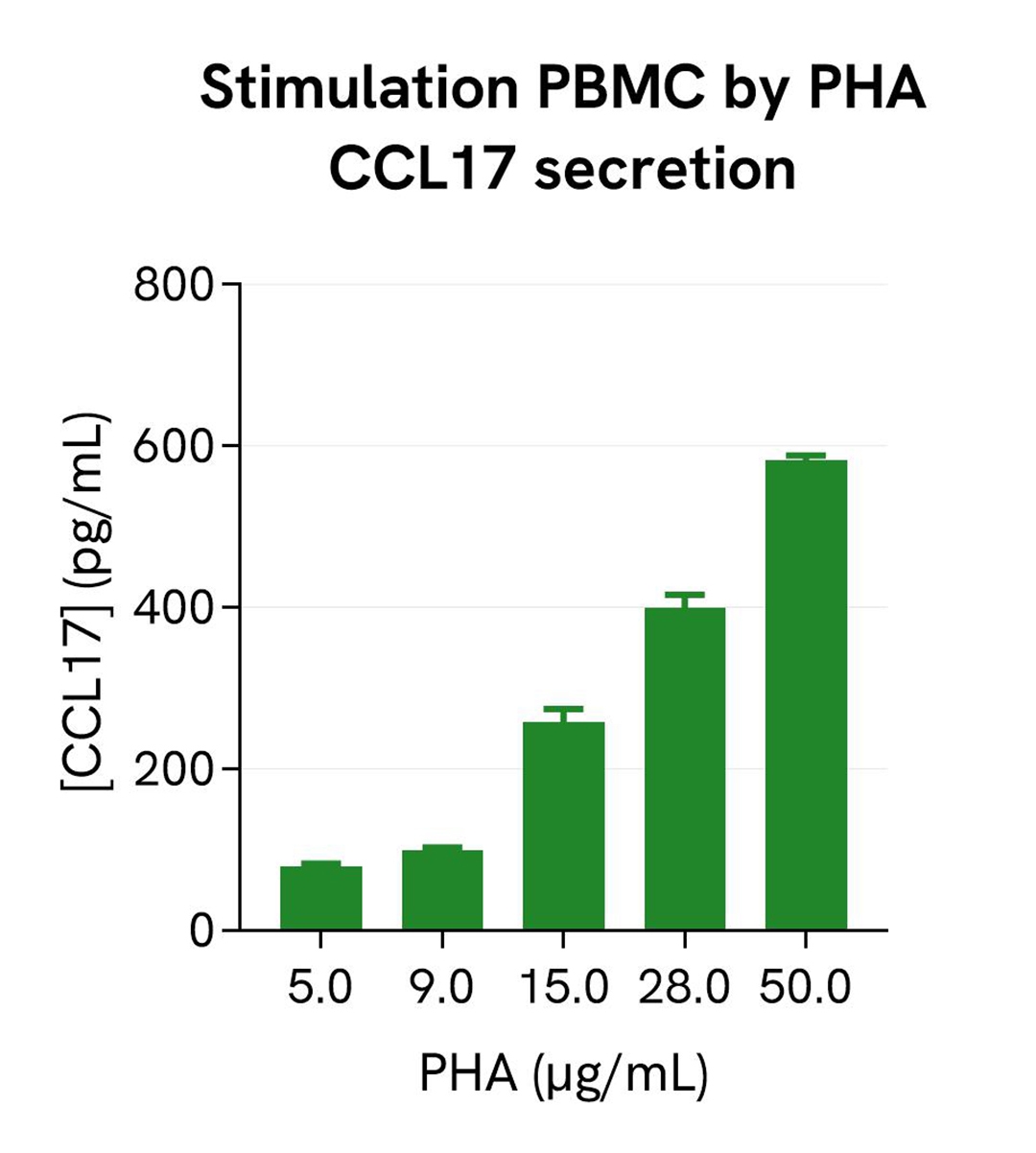
Dose response of PHA on PBMC
Human PBMCs (750,000 cells/well in RPMI) were stimulated with increasing concentrations of PHA for 24h, in a humidified atmosphere at 37°C and 5% CO2. After 24h of stimulation, the cell supernatants were collected. 16 µL of each sample were transferred into a white detection plate (384 wells, low volume) to measure the secreted CCL17 in each sample. Then 4 µL of the HTRF human CCL17 detection reagents were added to the wells. After 2h, the signals were recorded for every sample. The CCL17 concentration in each supernatant sample was extrapolated from the standard curve prepared in RPMI.
As expected, the CCL17 secretion increased with the PHA concentration, and reached a plateau (at around 30µg/mL).
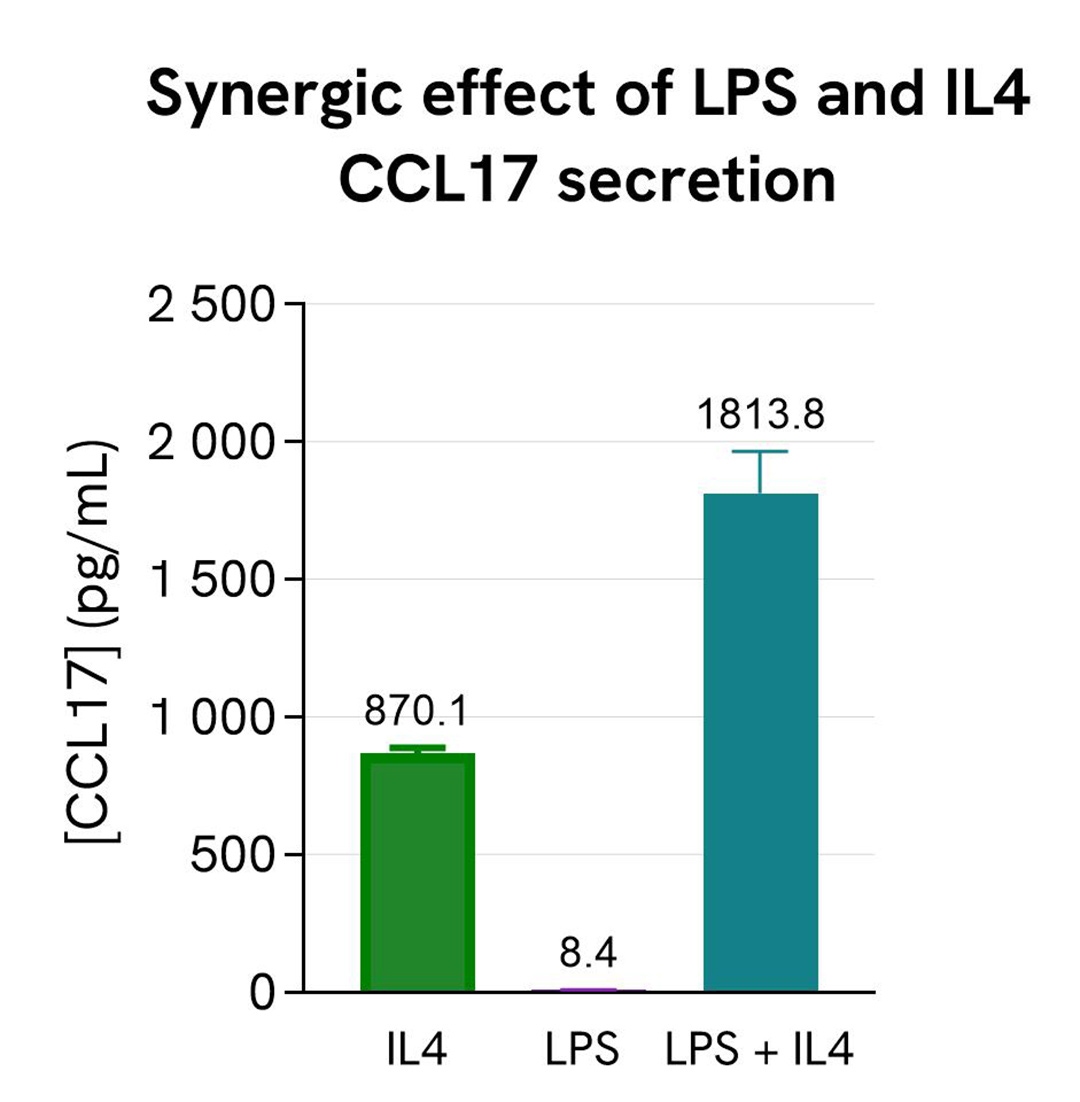
Synergistic effect of co-stimulation of IL4 and LPS
Human PBMCs (750,000 cells/well in RPMI) were stimulated with 20 ng/mL of IL4 or 2 µg/mL of LPS, or co-stimulated with both IL4 and LPS. The plate was placed in a humidified atmosphere at 37°C and 5% CO2.
After 24h of stimulation, the cell supernatants were collected.
16 µL of each sample were transferred into a white detection plate (384 wells, low volume) to measure the secreted CCL17 in each sample. Then 4 µL of the HTRF human CCL17 detection reagents were added to the wells. After 2h, the signals were recorded for every sample. The CCL17 concentration in each supernatant sample was interpolated from the standard curve prepared in RPMI.
IL4 stimulated the secretion of CCL17 in PBMCs, while LPS alone did not. However, there was a synergy effect from the co-stimulation of LPS and IL4, resulting in more secretion of CCL17 than with IL4 alone, in agreement with the literature (J. Immunol. (2017) 198 (9): 3426-3435).
Specifications
| Application |
Protein Quantification
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Dynamic Range |
21 - 5500 pg/mL
|
| Limit of Detection |
20 pg/mL
|
| Limit of Quantification |
35.3 pg/mL
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target |
CCL17
|
| Target Class |
Cytokines
|
| Target Species |
Human
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Inflammation
|
| Unit Size |
10,000 assay points
|
Resources
Are you looking for resources, click on the resource type to explore further.


How can we help you?
We are here to answer your questions.






























