
HTRF Human TREM2 (Full-Length) Detection Kit, 500 Assay Points
This HTRF kit enables cell-based quantitative detection of full-length TREM2.
| Feature | Specification |
|---|---|
| Application | 細胞シグナル伝達 |
| Sample Volume | 16 µL |
This HTRF kit enables cell-based quantitative detection of full-length TREM2.


Loading...
Product information
Overview
The kit is designed for the rapid detection of the full-length TREM2 receptor by using two antibodies that target distinct regions, the extracellular domain and the intracellular region, allowing detection before receptor shedding by ADAM10 and ADAM17 proteases. TREM2 (Triggering Receptor Expressed on Myeloid Cells 2) belongs to the TREM family of receptors, which regulate the activity of various immune cell types, including neutrophils, monocytes/macrophages, microglia, and dendritic cells.
HTRF assays offer many advantages over other technologies:
- Homogeneous add-and-read format
- No wash steps
- Low background
- Straightforward miniaturization from 96- or 384-well microplates to high density assay formats such as 384-well low volume and 1536-well plates
- Stable signal, providing flexibility in readout time or size of assays
How it works
HTRF TREM2 full-length assay principle
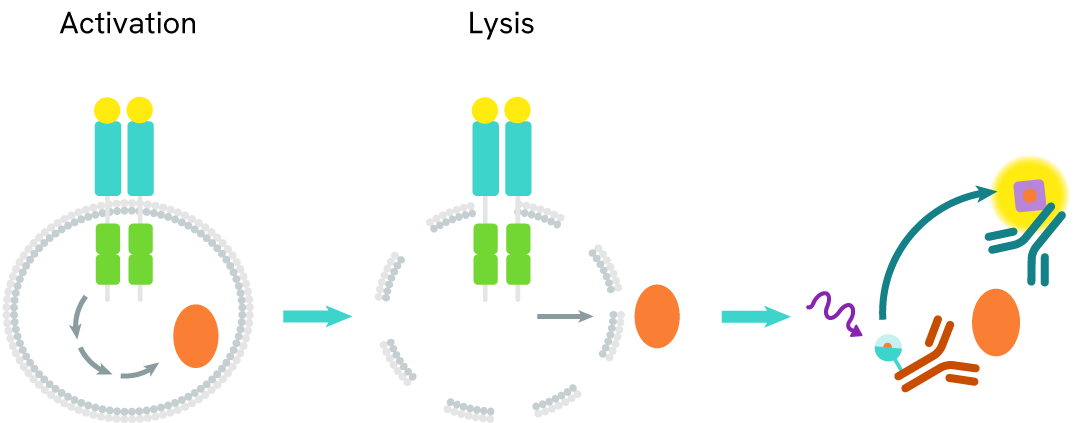
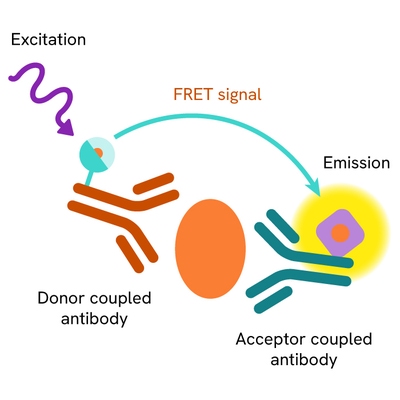
The TREM2 full-length assay measures full-length TREM2 levels in cells. Unlike Western Blot, the assay is entirely plate-based and does not require gels, electrophoresis, or transfer. It uses two antibodies: one labeled with a donor fluorophore and the other with an acceptor. Both antibodies are highly specific for distinct epitopes on the protein.
In the presence of full-length TREM2, an immune complex forms involving both labeled antibodies, bringing the donor fluorophore into close proximity to the acceptor and generating a FRET signal. The signal intensity is directly proportional to the concentration of full-length TREM2 in the sample.

HTRF TREM2 full-length two-plate assay protocol
The two-plate protocol involves culturing cells in a 96-well plate before lysis, then transferring lysates into a 384-well low volume detection plate before the addition of TREM2 Full-Length HTRF detection reagents. This protocol allows the cells' viability and confluence to be monitored.
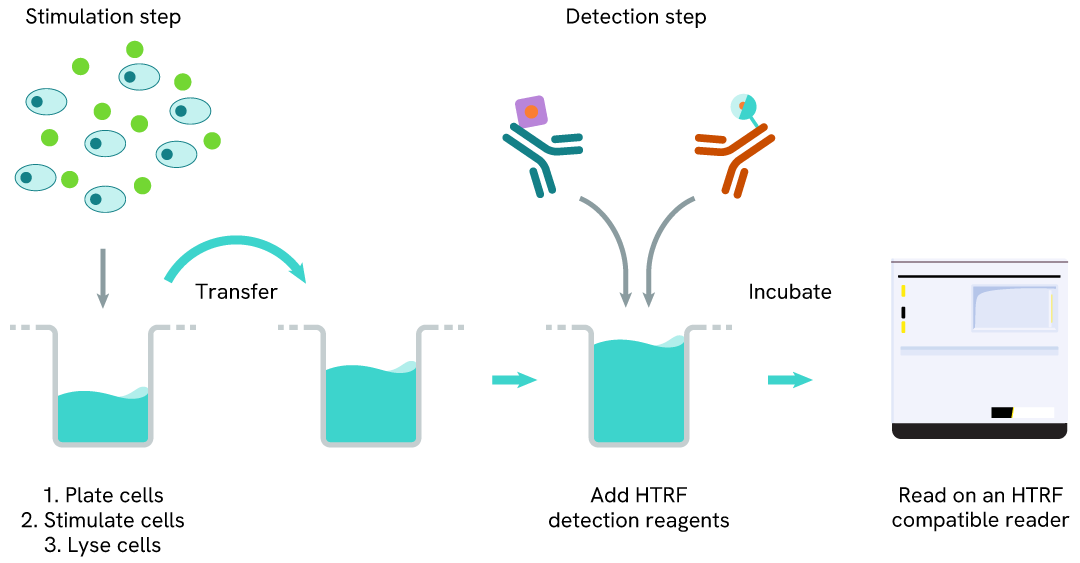
HTRF TREM2 full-length one-plate assay protocol
Detection of TREM2 full-length with HTRF reagents can be performed in a single plate used for culturing, stimulation, and lysis. No washing steps are required. This HTS designed protocol allows miniaturization while maintaining robust HTRF quality.
Assay validation
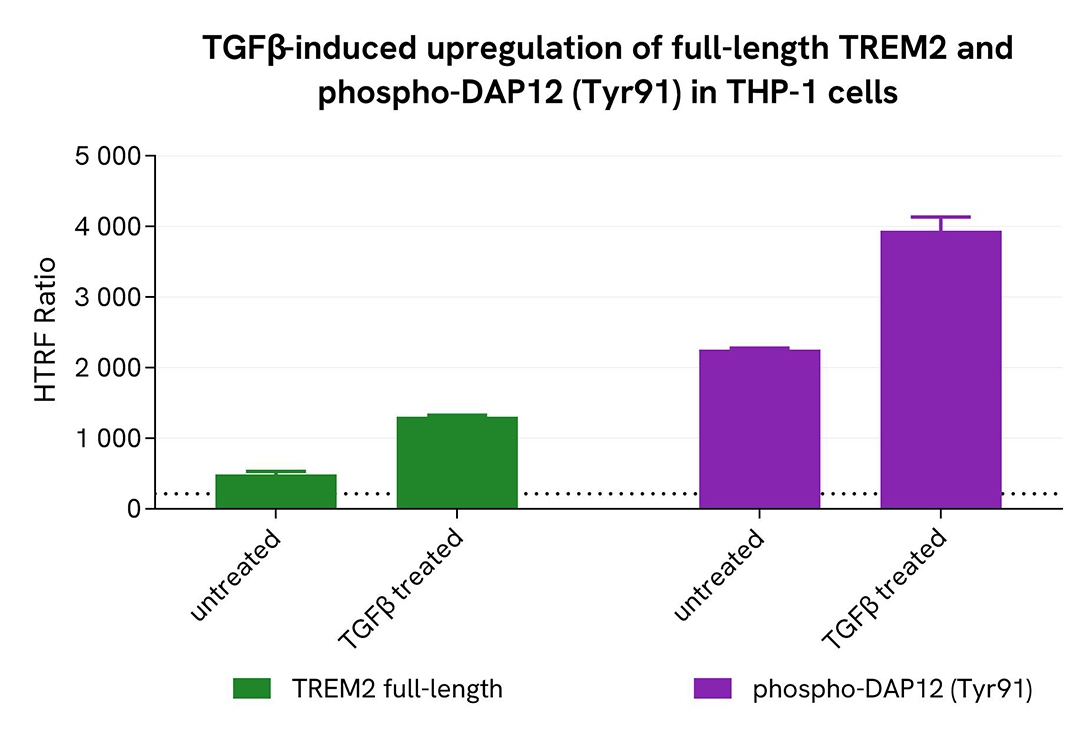
Induction of full-length TREM2 and phospho-DAP12 (Tyr91) in an endogenous cellular model
THP-1 cells were plated in a 96-well microplate (100,000 cells/well) and treated with 35 ng/mL TGFβ for 20 hours at 37°C. After incubation, the supernatant was removed, and cells were lysed with 50 µL of supplemented lysis buffer #3 (1x) for 30 minutes at room temperature under gentle shaking.
Next, 16 µL of cell lysate were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of premixed HTRF detection reagents. The HTRF signal was recorded after 3 hours of incubation.
As expected, TGFβ induced activation of the TREM2 pathway, resulting in an increase in full-length TREM2 and Phospho-DAP12 (Tyr91) levels.
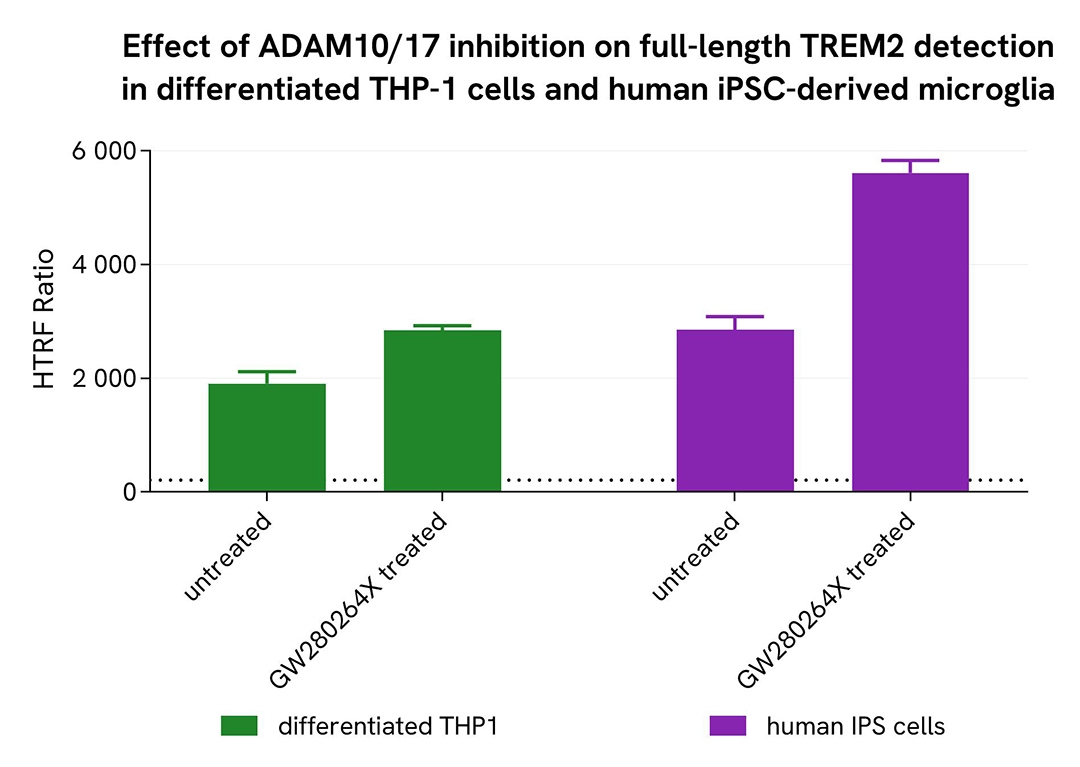
Shedding inhibition in differentiated THP-1 cells and microglia derived from human iPSCs
THP-1 cells were seeded at 100,000 cells per well in a 96-well microplate and treated for 48 hours with 100 nM PMA at 37°C, 5% CO2. Microglia differentiated from human iPSCs (iCell® Microglia, Fujifilm Cellular Dynamics) were seeded at 50,000 cells per well in a 96-well microplate for 72 hours under the same conditions.
After a centrifugation step at 200 × g for 3 minutes, the supernatant was removed, and cells were lysed with supplemented lysis buffer #3 (1x) for 30 minutes at room temperature under gentle shaking.
Next, 16 µL of lysate were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of premixed HTRF detection reagents. The HTRF signal was recorded after 3 hours of incubation.
As expected, the selective ADAM10/17 inhibitor GW280264X inhibited shedding and led to an increase in full-length TREM2 detection.
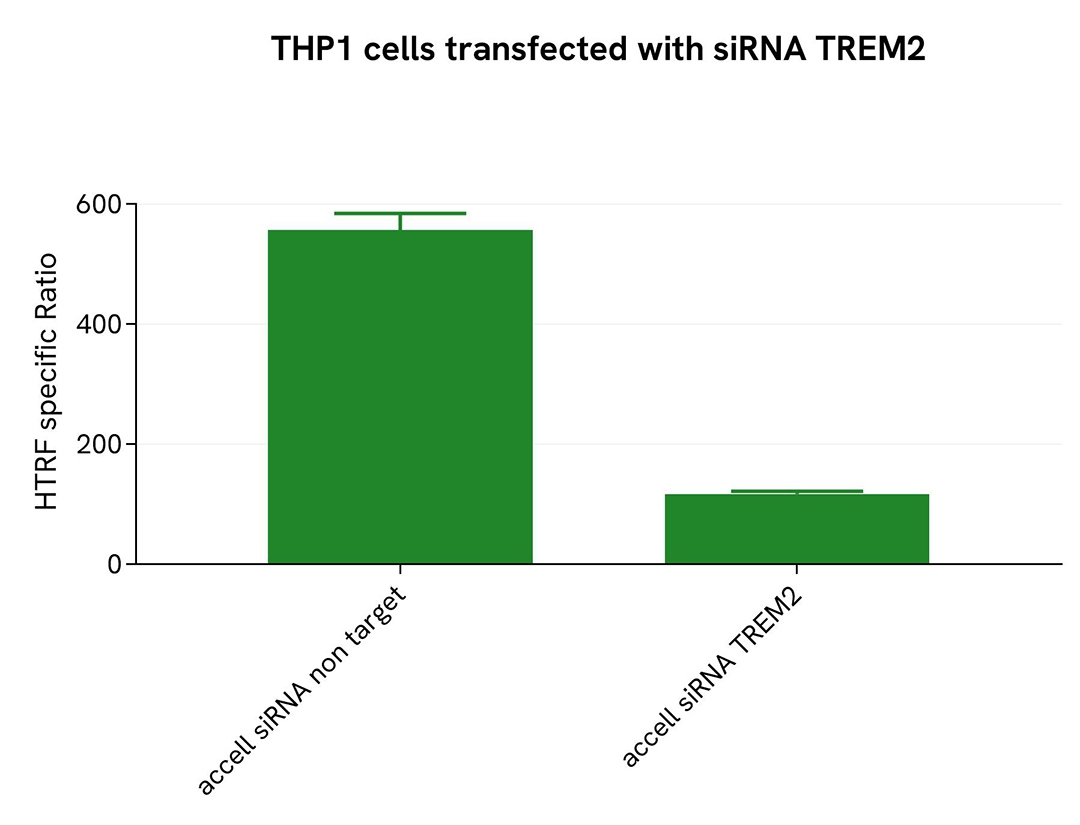
Validation of TREM2 full-length assay selectivity using Accell siRNA
THP-1 cells were plated in a 96-well microplate (25,000 cells/well) and transfected with Accell TREM2 siRNA or a negative control. After a 72-hour incubation, 100 nM PMA were added for 48 hours before cells were lysed with lysis buffer #3 (1x) for 30 minutes at room temperature under gentle shaking.
For detection, 16 µL of lysate were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of premixed HTRF detection reagents. The HTRF signal was recorded after 3 hours of incubation.
Transfection with TREM2 siRNA resulted in a significant decrease (79%) in protein detection compared to the negative control (non-targeting siRNA), demonstrating the selectivity of the HTRF TREM2 Full-Length assay.
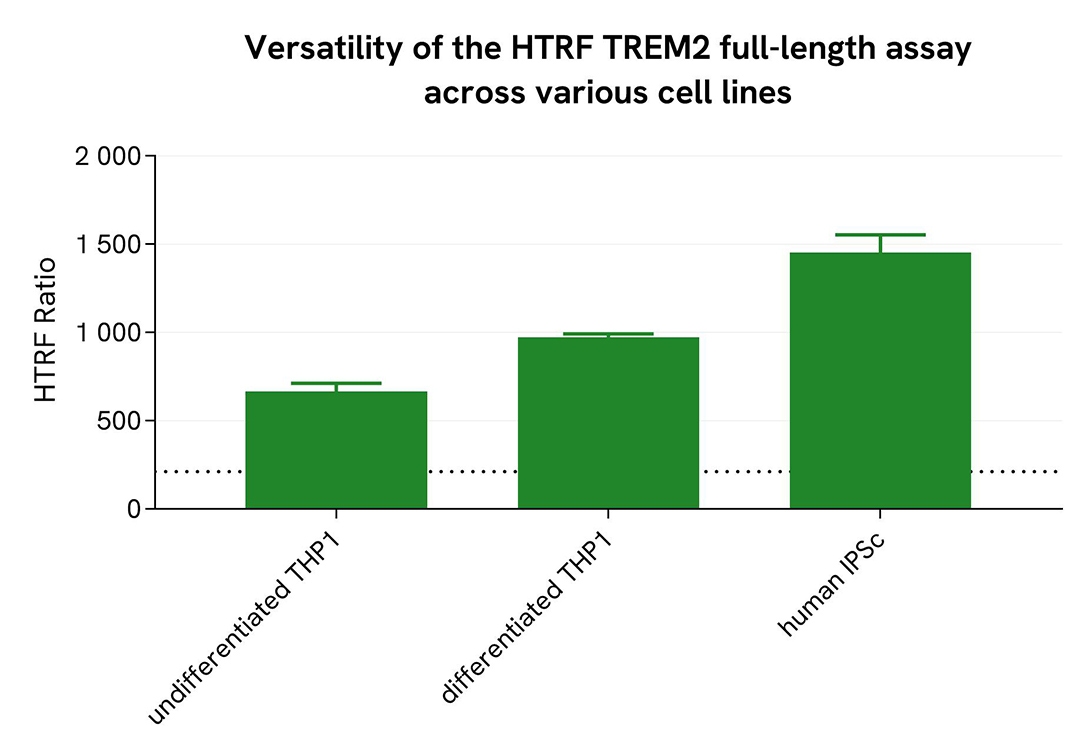
Assessment of full-length TREM2 levels in various cell lines
Undifferentiated and differentiated THP-1 cells (treated with 100 nM PMA for 20 hours) and microglia derived from human iPSCs (iCell® Microglia, Fujifilm Cellular Dynamics) were seeded at 25,000 cells per well in a 96-well microplate for 20 hours. After incubation, the supernatant was removed, and cells were lysed with lysis buffer #3 for 30 minutes at room temperature under gentle shaking, following the protocol for adherent or suspension cells.
Next, 16 µL of sample were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of premixed HTRF detection reagents. The HTRF signal was recorded after 3 hours of incubation.
The HTRF full-length TREM2 assay successfully detected full-length TREM2 across multiple cellular models, revealing differential expression levels.
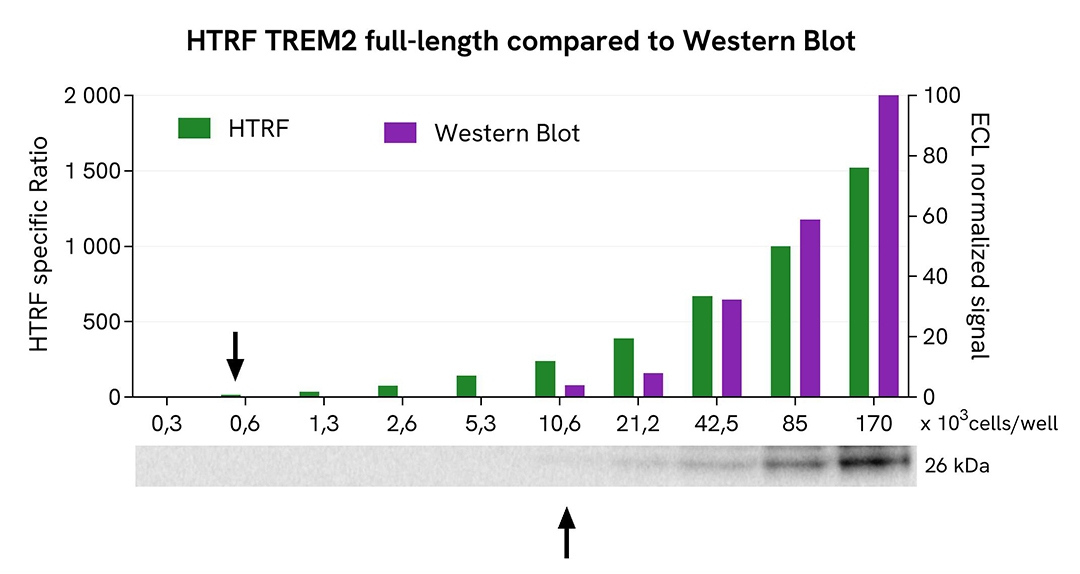
HTRF TREM2 full-length assay compared to Western Blot
THP-1 cells were grown in a T175 flask in complete culture medium at 37°C, 5% CO2 until 80% confluence was reached. The cells were then treated with 35 ng/mL TGFβ for 20 hours and lysed with 3 mL of supplemented lysis buffer #3 (1x) for 30 minutes at room temperature under gentle shaking.
Serial dilutions of the cell lysate were prepared using supplemented lysis buffer. Then 13 µL of each dilution were transferred into a low-volume white microplate, followed by the addition of 3 µL of supplemented lysis buffer #3 (1x) to ensure equal volume across techniques. Subsequently, 4 µL of HTRF TREM2 Full-Length detection reagents were added. Equal amounts of lysate were used for a side-by-side comparison between HTRF and Western Blot.
Using the HTRF TREM2 Full-Length assay, 664 cells per well were sufficient to detect a significant signal, whereas 10,625 cells were required to obtain a minimal chemiluminescent signal using Western Blot. Under these conditions, the HTRF TREM2 Full-Length assay was 16 times more sensitive than Western Blot.
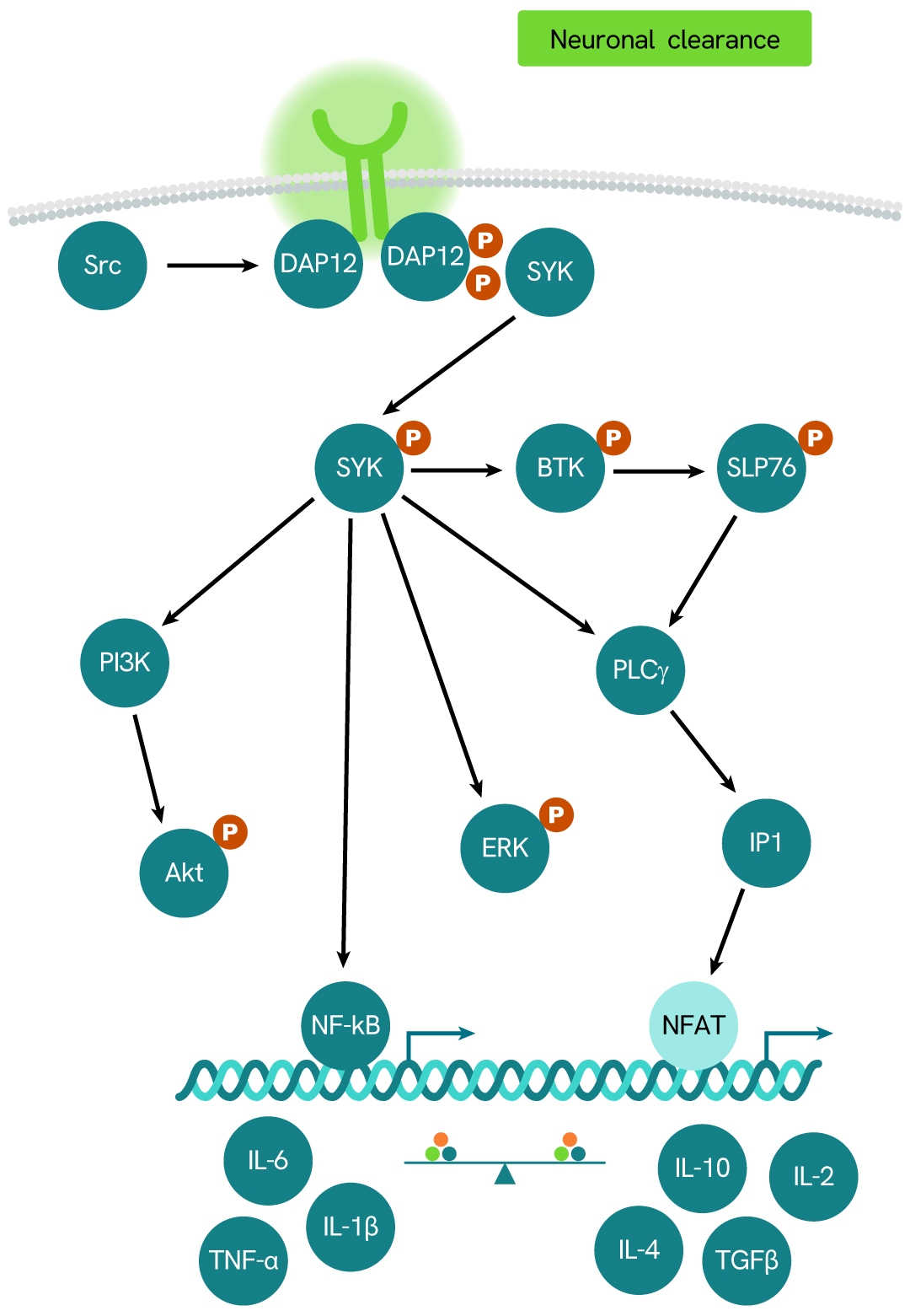
Simplified pathway
TREM2 signaling pathway
TREM2 (Triggering Receptor Expressed on Myeloid Cells 2) is a member of the TREM family of receptors, which regulate the activity of various immune cell types, including neutrophils, monocytes/macrophages, microglia, and dendritic cells. TREM2 requires the adaptor protein DAP12 for downstream signaling.
The receptor undergoes proteolytic cleavage by ADAM10 and ADAM17, a process known as shedding. This cleavage releases the ectodomain - the extracellular portion of the receptor - into the surrounding environment as soluble TREM2 (sTREM2), which has been detected in human cerebrospinal fluid (CSF). Elevated levels of sTREM2 have been observed in patients with multiple sclerosis and other inflammatory neurological conditions compared to those without inflammatory neurologic disorders.
Specifications
| Application |
Cell Signaling
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 1
Lysis Buffer 3
Lysis Buffer 4
|
| Molecular Modification |
Total
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target |
TREM2
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Neuroinflammation
Neuroscience
|
| Unit Size |
500 assay points
|
Resources
Are you looking for resources, click on the resource type to explore further.
Loading...


How can we help you?
We are here to answer your questions.






























