
HTRF Human Total SKP2 Detection Kit, 10,000 assay points
This HTRF kit allows the cell-based quantitative detection of Total SKP2.
| Feature | Specification |
|---|---|
| Application | 細胞シグナル伝達 |
| Sample Volume | 16 µL |
This HTRF kit allows the cell-based quantitative detection of Total SKP2.


Loading...
Product information
Overview
S-phase kinase-associated protein 2 (SKP2), also known as FBXL1, is a prominent member of the family of F-box proteins. It functions as an E3 ligase mainly through the formation of an SCF-SKP2 complex. SKP2 is well known for its capacity to regulate the cell cycle. It can ubiquitinate various cell cycle regulators and induce their hydrolysis, including CKIs p21, p27, and p57, as well as the cell cycle protein E (cyclin E) and the cell cycle regulator c-Myc. As a driver of oncogenesis and drug resistance, SKP2 is an attractive target for small-molecule inhibitors and protein degradation strategies aimed at restoring cell cycle checkpoints.
HTRF assays offer many advantages over other technologies:
- Homogeneous add-and-read format
- No wash steps
- Low background
- Straightforward miniaturization from 96- or 384-well microplates to high density assay formats such as 384-well low volume and 1536-well plates
- Stable signal, providing flexibility in readout time or size of assays
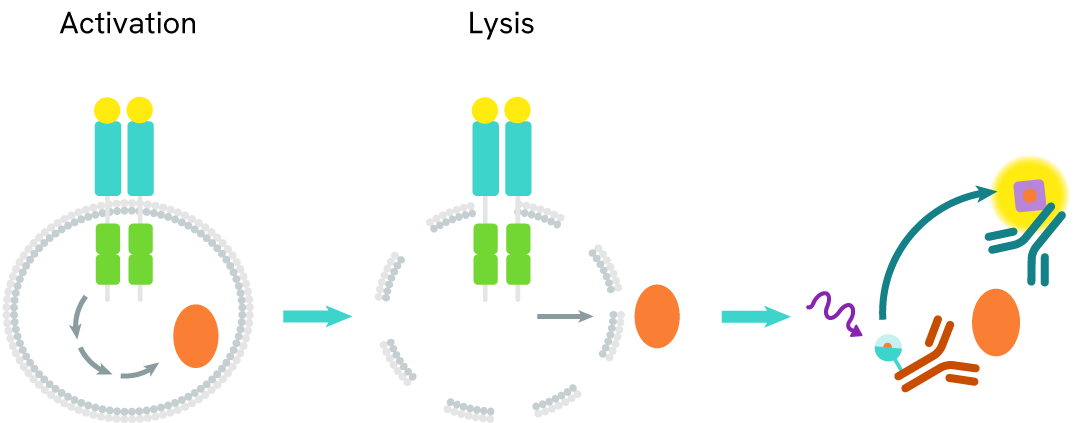
How it works
Total SKP2 assay principle
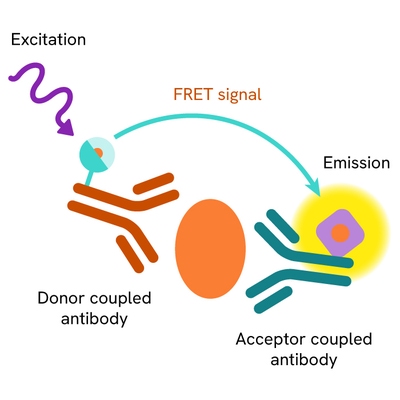
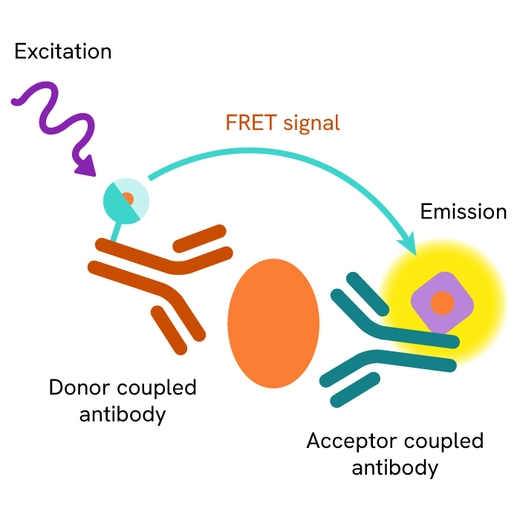
The Total SKP2 assay measures SKP2 levels in cells. Unlike Western Blot, the assay is entirely plate-based and does not require gels, electrophoresis, or transfer. The assay uses 2 antibodies, one labeled with a donor fluorophore and the other with an acceptor. Both antibodies are highly specific for a distinct epitope on the protein.
In the presence of SKP2 this enables an immune-complex formation involving both labeled antibodies, and which brings the donor fluorophore into close proximity to the acceptor, thereby generating a FRET signal. Its intensity is directly proportional to the concentration of total protein present in the sample and provides a means of assessing the protein's phosphorylation state under a no-wash assay format.

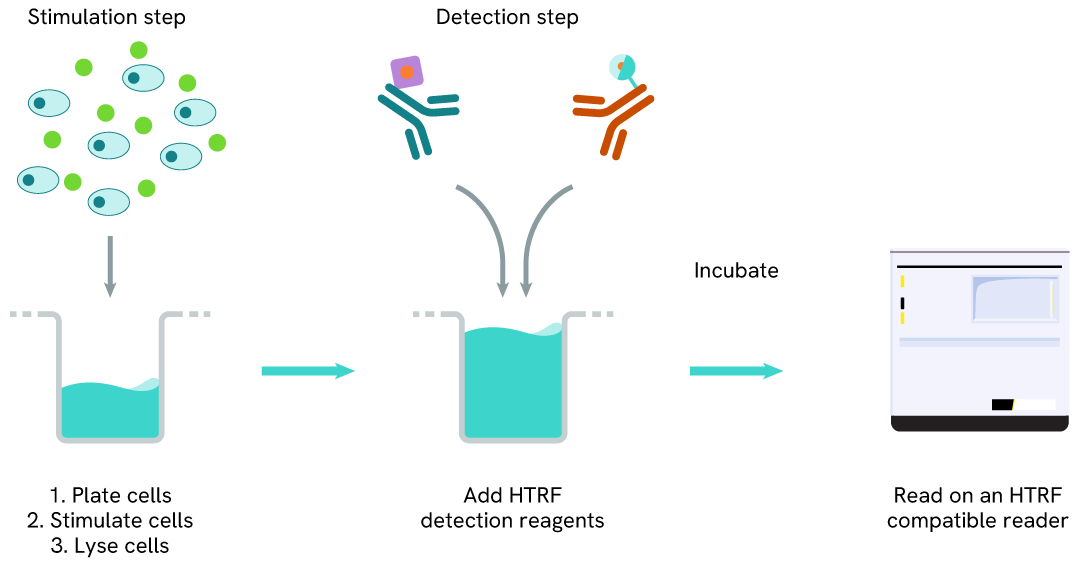
Total SKP2 two-plate assay protocol
The two-plate protocol involves culturing cells in a 96-well plate before lysis, then transferring lysates into a 384-well low volume detection plate before the addition of Total SKP2 HTRF detection reagents. This protocol allows the cells' viability and confluence to be monitored.
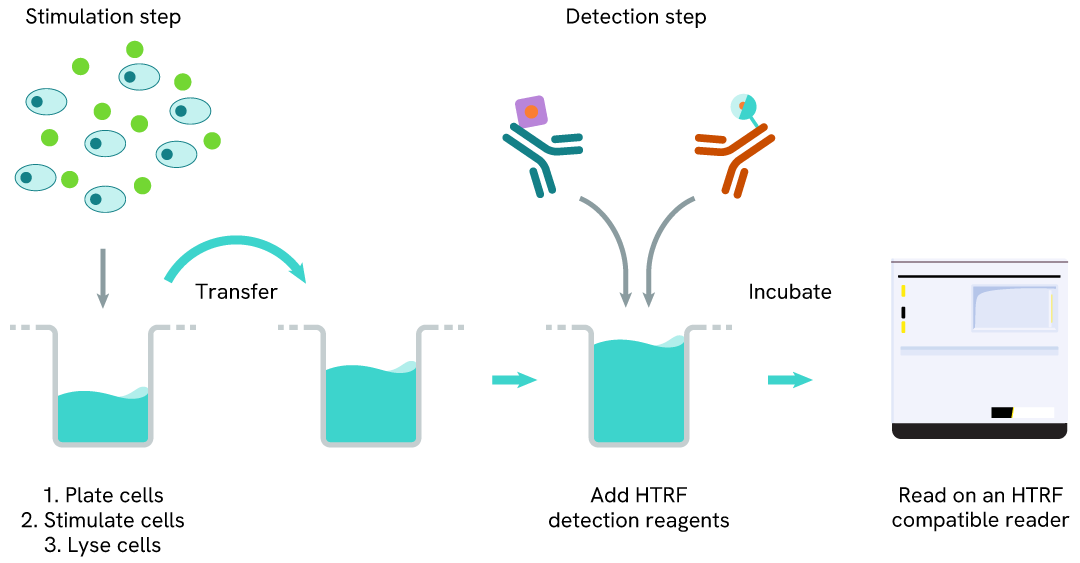
Total SKP2 one-plate assay protocol
Detection of Total SKP2 with HTRF reagents can be performed in a single plate used for culturing, stimulation, and lysis. No washing steps are required. This HTS designed protocol allows miniaturization while maintaining robust HTRF quality.
Assay validation
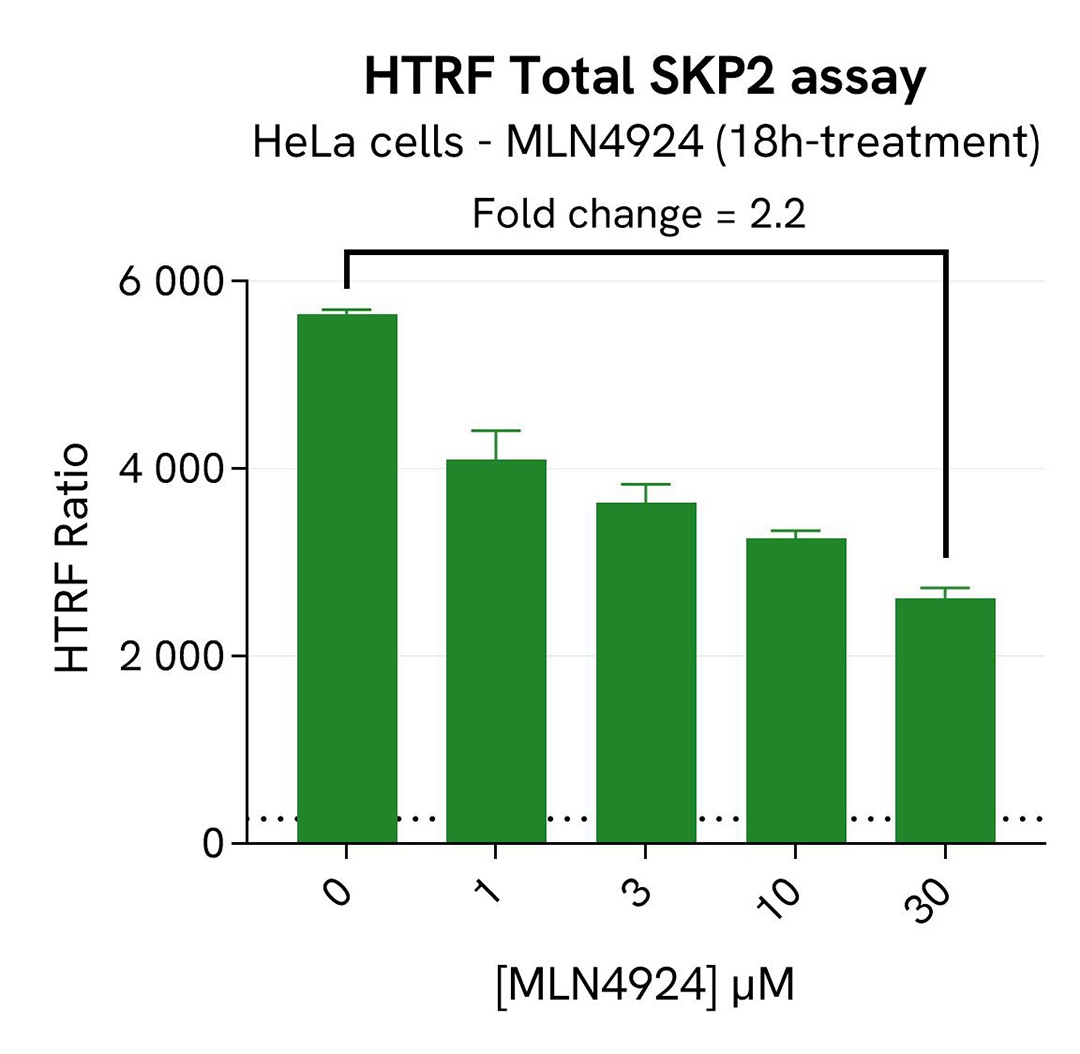
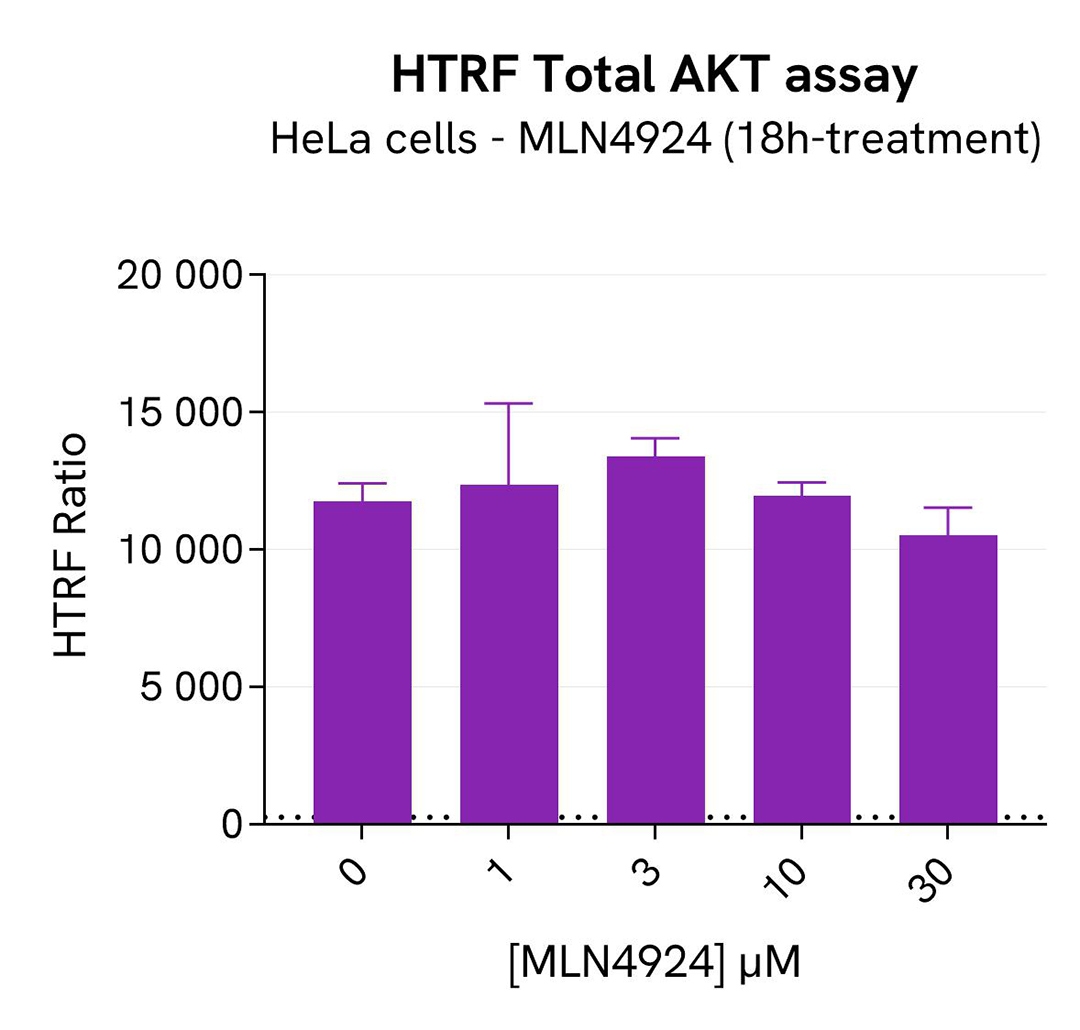
SKP2 degradation induced by MLN4924 in HeLa cells
HeLa cells were seeded in a 96-well culture-treated plate (40,000 cells/well) in complete culture medium and incubated overnight at 37°C, 5% CO2. The cells were then treated for 18h with increasing concentrations of MLN4924, an inhibitor of Cullin neddylation that promotesSKP2 degradation. After treatment, the cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at RT under gentle shaking.
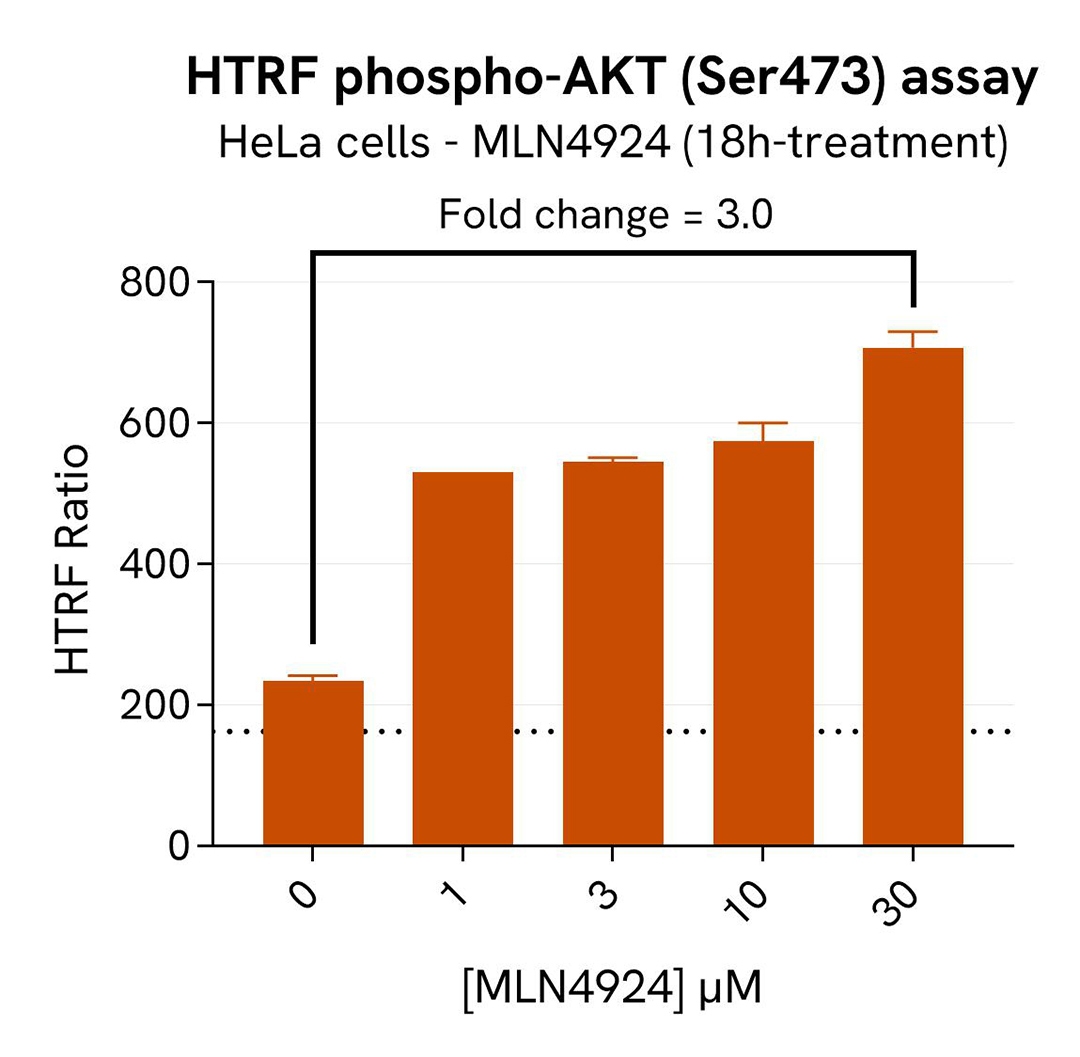
For the detection step, 16 µL of cell lysate were transferred into a 384-well low volume white microplate and supplemented with 4 µL of the HTRF Total SKP2detection reagents. After overnight incubation, the HTRF signal was recorded with an Envision Nexus reader . Additionally, AKT has been described as participating in SKP2 regulation by protecting it from Cdh1-mediated ubiquitination degradation. The activation of AKT under SKP2 inhibition conditions was monitored in the same experiment with the HTRF Total AKT or HTRF Phospho-AKT (Ser473) Detection Kits (Revvity, #64NKTPEG & #64AKSPEG). The corresponding HTRF signals were recorded with an Envision Nexus reader after 4h incubation for Phospho-AKT (Ser473) and overnight incubation for Total AKT.
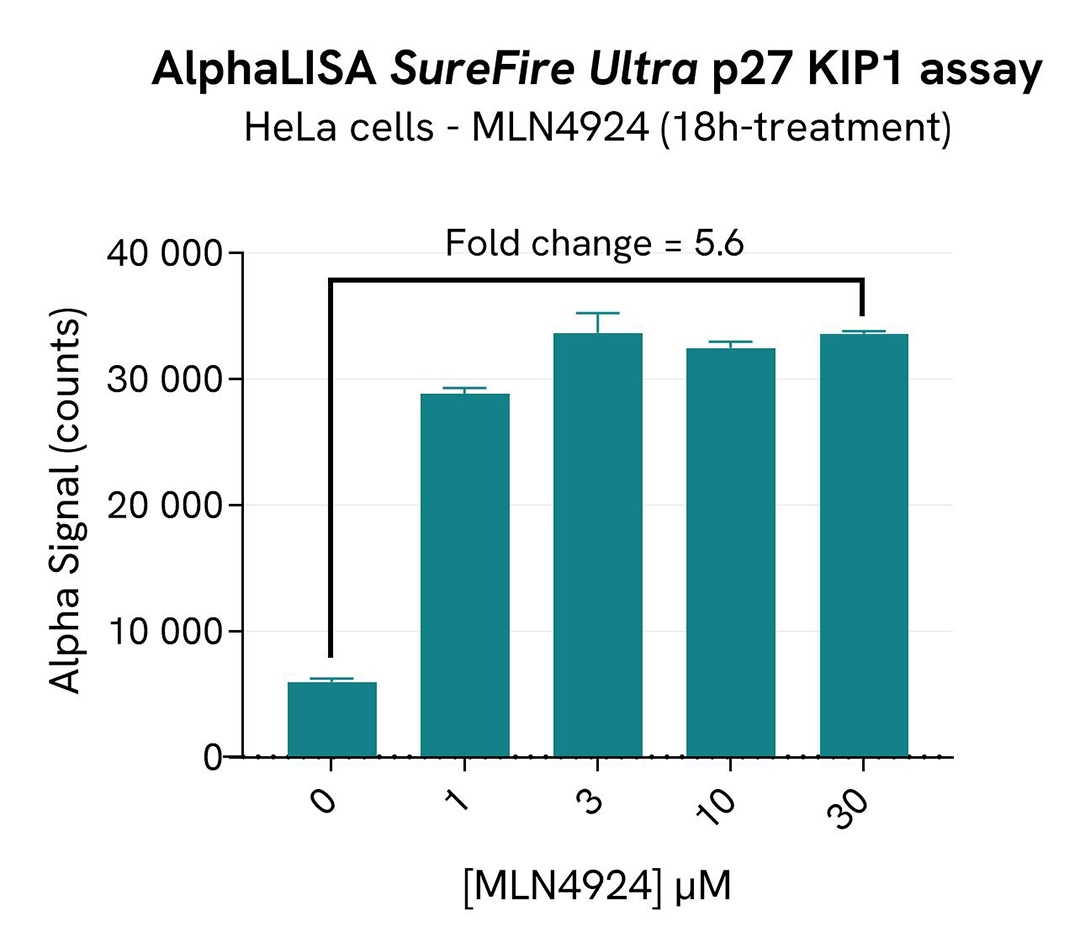
In parallel, the effect of SKP2 inhibition on p27 (ubiquitinylation target of SKP2) were monitored with the AlphaLISA SureFire Ultra Human and Mouse p27 KIP1 Total Detection Kit (#ALSU-TP27K). 10 µL of cell lysate were transferred into an AlphaPlate-384 microplate (Revvity, #6005350) and 10 µL of the corresponding detection reagents were added. After a 2h incubation, the Alpha signal was recorded with an Envision Nexus reader.
Finally, cell viability was assessed with the ATPlite 1step Kit (Revvity, #6016736/1/9). 5 µL of the same cell lysate were transferred into an HTRF 96-well low volume white plate, and 25 µL of ATPlite 1step detection reagent were added. After a 10-min incubation, the luminescence signal was measured with an Envision Nexus reader.
As expected, the Cullin neddylation inhibitor MLN4924 induced SKP2 degradation and an increased p27 KIP1 level as well as phosphorylation of AKT on Ser473, without affecting the Total AKT signal. In addition, MLN4924 did not induce cytotoxic effects, as measured by the cell viability indicator ATPLite (data not shown).
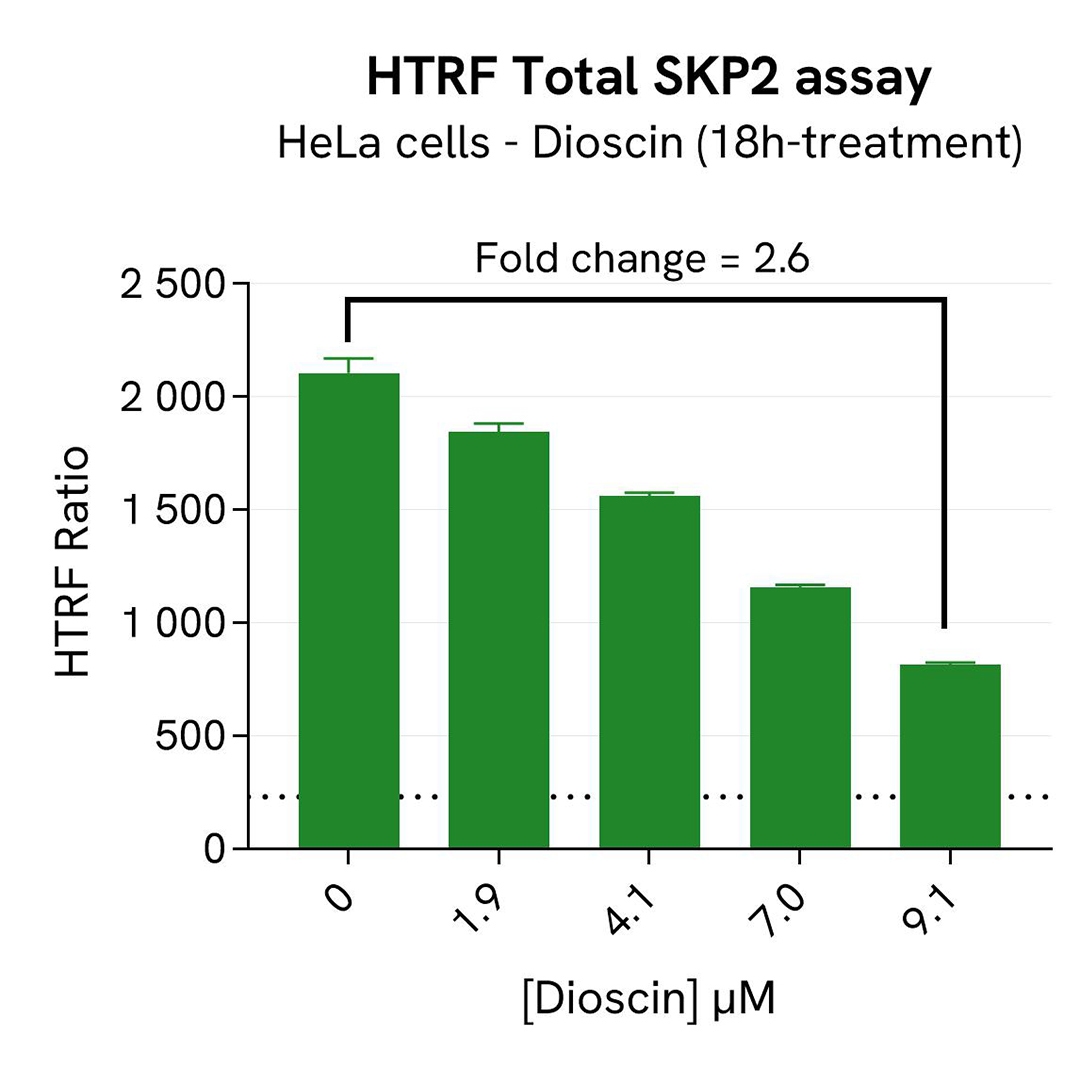
SKP2 degradation induced by Dioscin in HeLa cells
HeLa cells were seeded in a 96-well culture-treated plate (40,000 cells/well) in complete culture medium, and incubated overnight at 37°C, 5% CO2. The cells were treated for 18h with increasing concentrations of Dioscin, an inhibitor of SKP2 that induces its ubiquitination and its degradation by APC.
After treatment, the cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at RT under gentle shaking. For the detection step, 16 µL of cell lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF Total SKP2 detection reagents were added. After overnight incubation, the HTRF signal was recorded with an Envision Nexus reader.
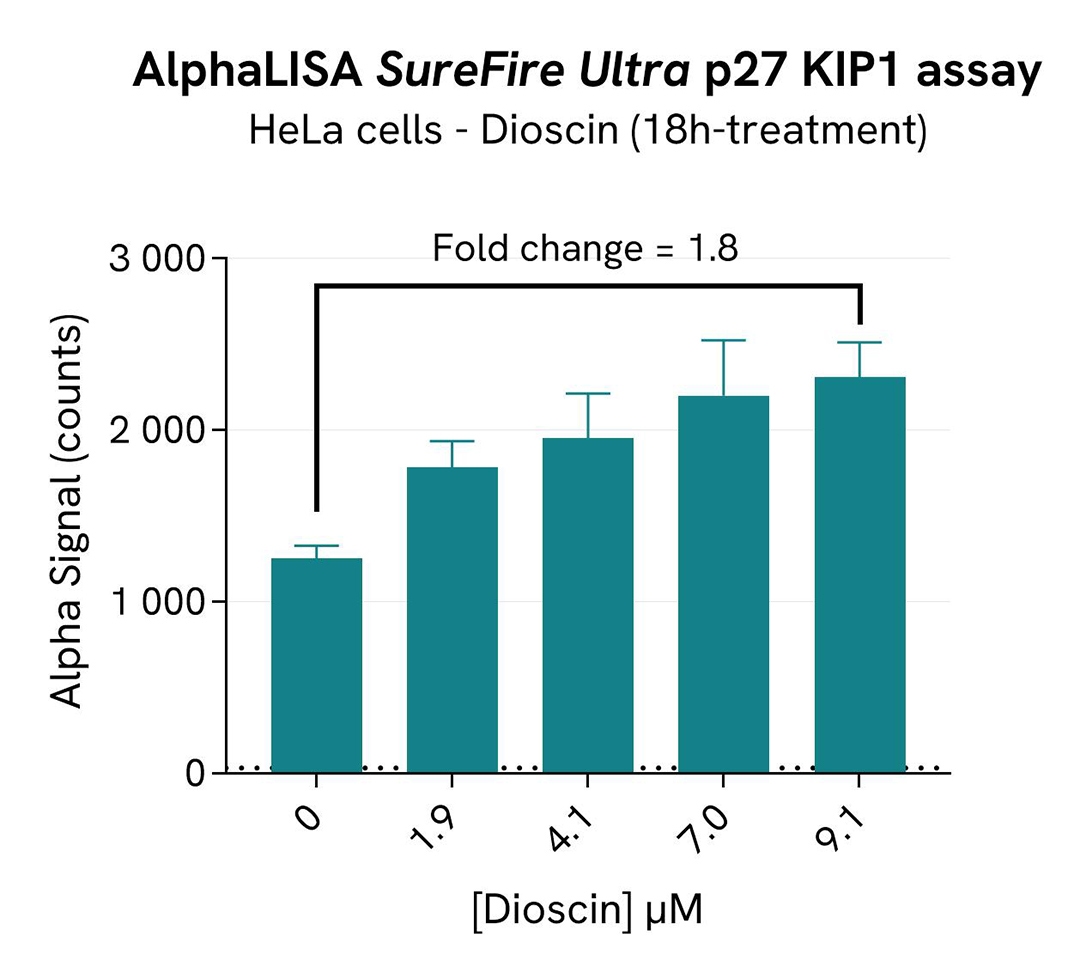
In parallel, the effects of SKP2 inhibition on p27 were monitored using the AlphaLISA SureFire Ultra Human and Mouse p27 KIP1 Total Detection Kit (#ALSU-TP27K).10 µL of cell lysate were transferred into an AlphaPlate-384 microplate (Revvity, #6005350) and 10 µL of the corresponding detection reagents were added. After a 2h incubation,Tte Alpha signal was recorded with an Envision Nexus reader.
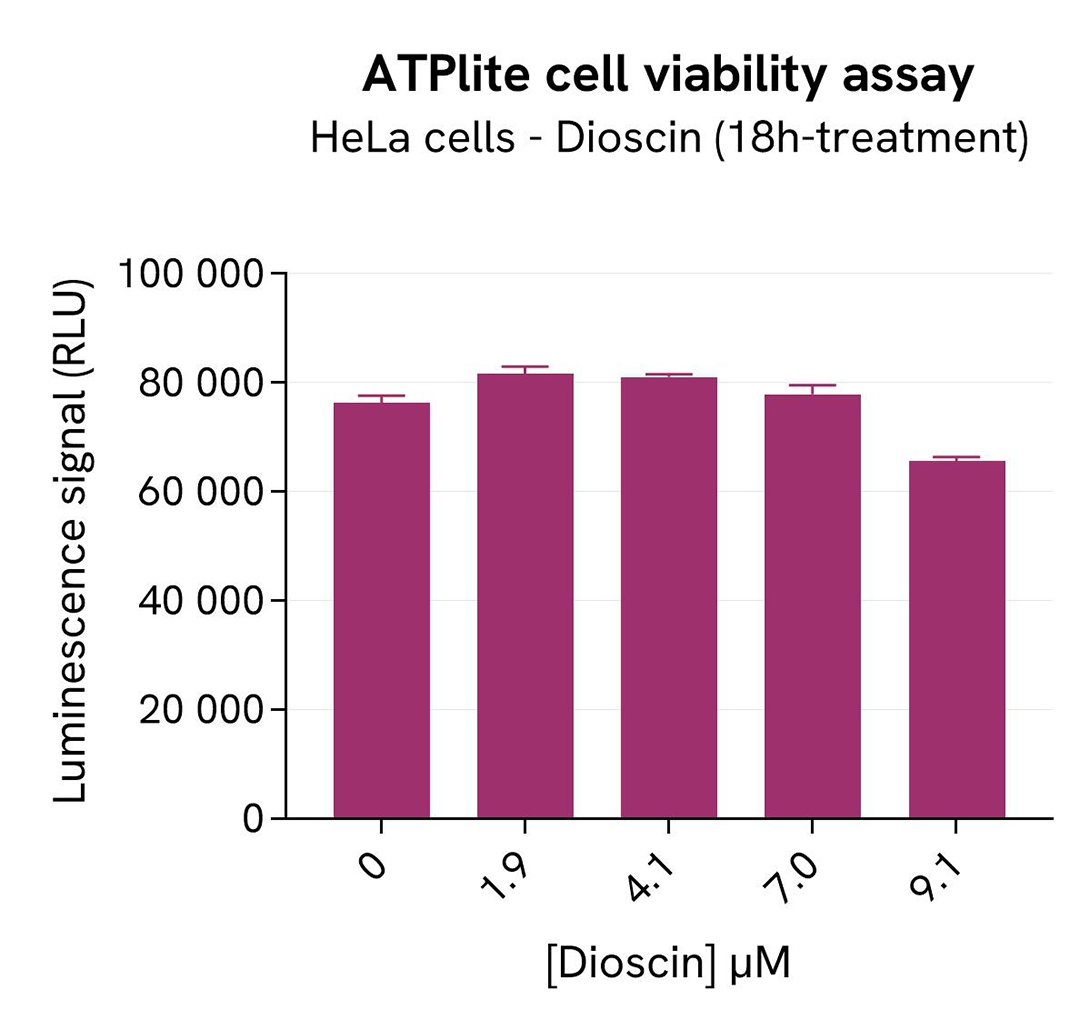
Finally, cell viability was assessed alongside the previous assays with the ATPlite 1step Kit (Revvity, #6016736/1/9). 5 µL of the same cell lysate were transferred into an HTRF 96-well low volume white plate, and 25 µL of ATPlite 1step detection reagent. After a 10-min incubation, the luminescence signal was measured with an Envision Nexus reader.
As expected, the SKP2 inhibitor Dioscin induced SKP2 degradation, which increased the p27 KIP1 level. In addition, Dioscin did not induce cytotoxic effects, as measured by the cell viability indicator ATPLite.
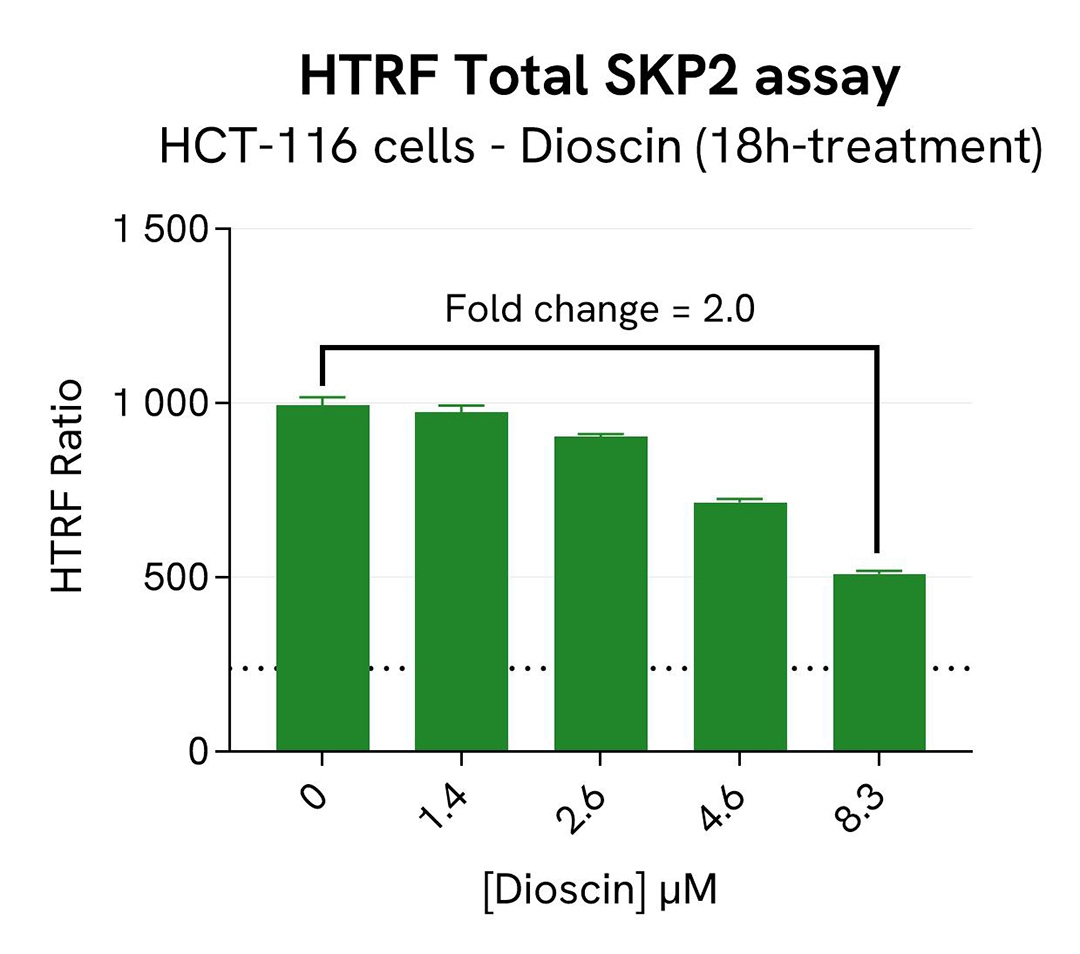
SKP2 degradation induced by Dioscin in HCT-116 cells
HCT-116 cells were seeded in a 96-well culture-treated plate (20,000 cells/well) in complete culture medium, and incubated overnight at 37°C, 5% CO2. The cells were treated for 18h with increasing concentrations of Dioscin, an inhibitor of SKP2 that induces its ubiquitination and its degradation by APC. After treatment, the cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at RT under gentle shaking.
For the detection step, 16 µL of cell lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF Total SKP2 detection reagents were added. After an overnight incubation, the HTRF signal was recorded with an Envision Nexus reader.
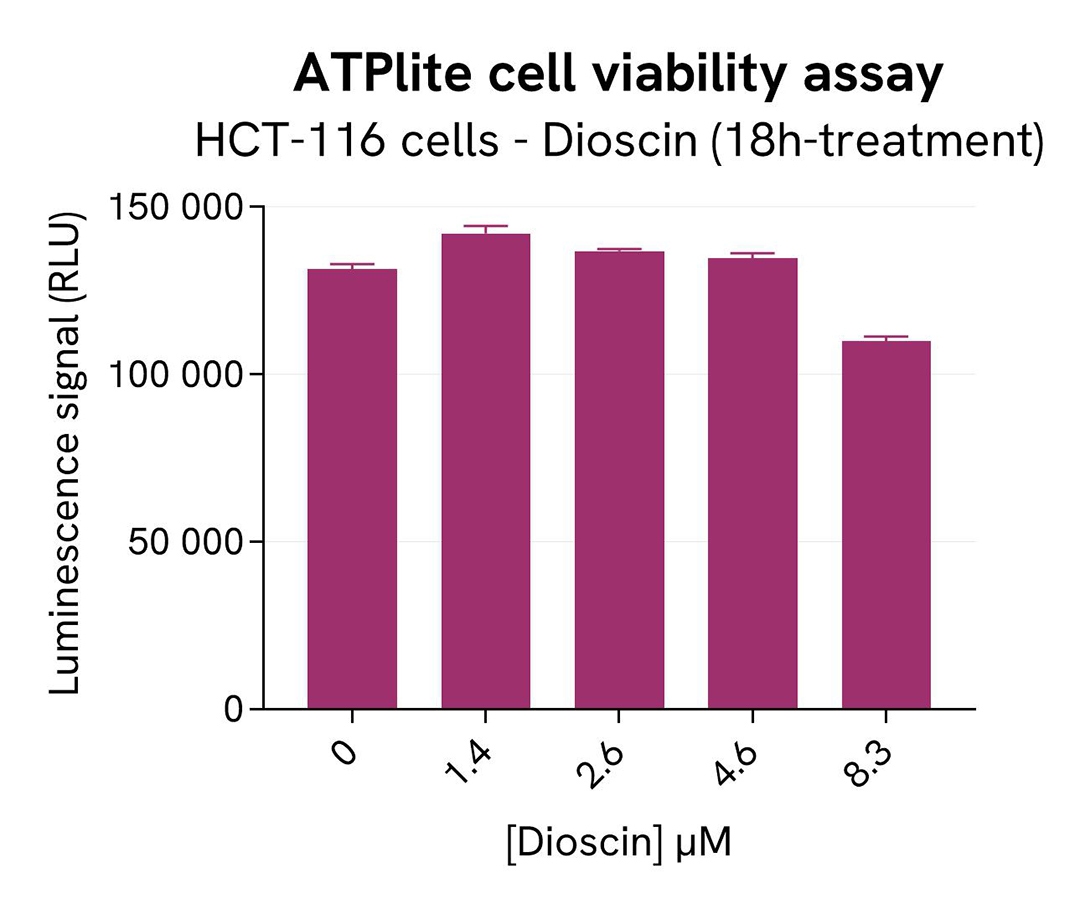
In parallel, cell viability was assessed with the ATPlite 1step Kit (Revvity, #6016736/1/9). 5 µL of the same cell lysate were transferred into an HTRF 96-well low volume white plate, and 25 µL of ATPlite 1step detection reagent were added. After a 10-min incubation, the luminescence signal was measured with an Envision Nexus reader.
As expected, the SKP2 inhibitor Dioscin induced SKP2 degradation. In addition, Dioscin did not induce cytotoxic effects, as measured by the cell viability indicator ATPLite.
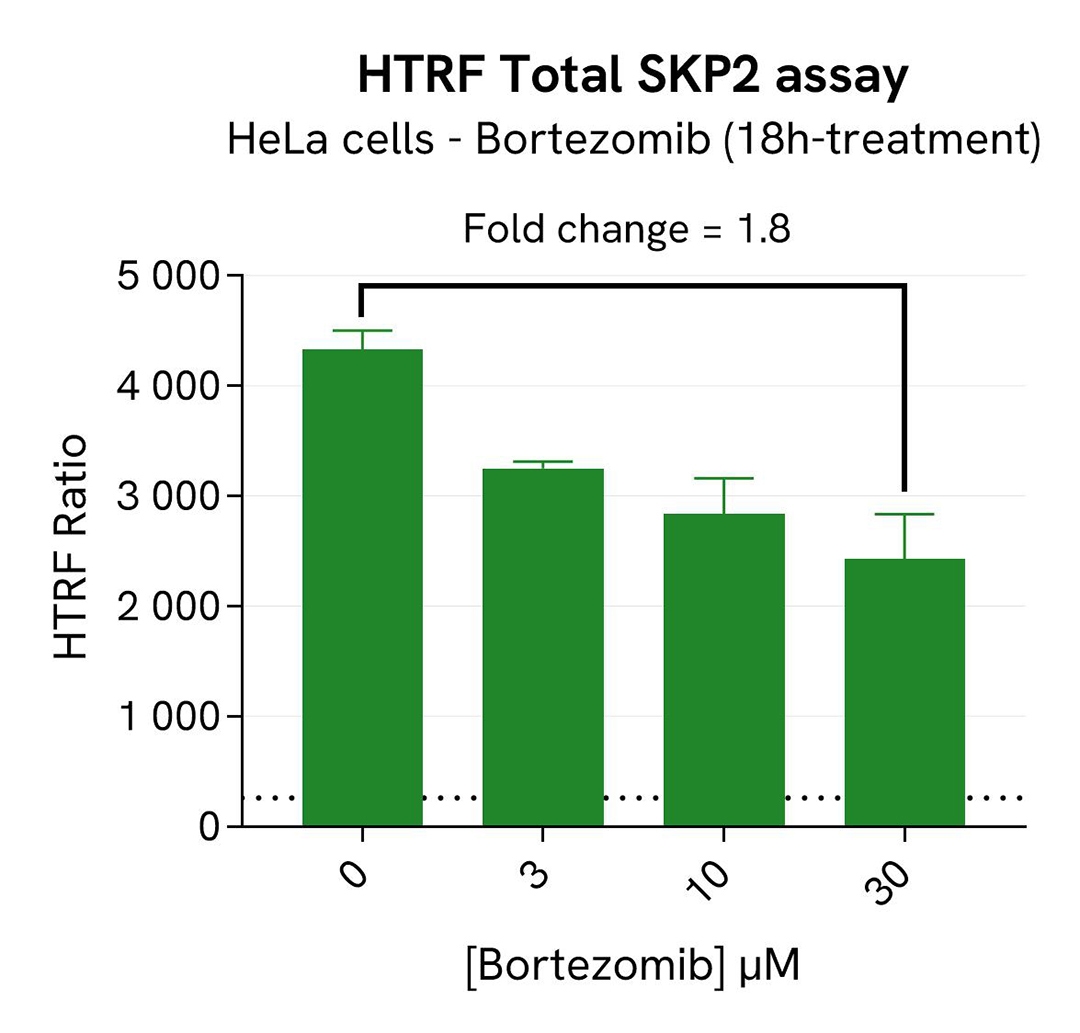
SKP2 expression decrease induced by Bortezomib in HeLa cells
HeLa cells were seeded in a 96-well culture-treated plate (40,000 cells/well) in complete culture medium, and incubated overnight at 37°C, 5% CO2. The cells were treated for 18h with increasing concentrations of Bortezomib, an inhibitor of the 26S proteasome that suppresses the expression of SKP2.
After treatment, the cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at RT under gentle shaking. For the detection step, 16 µL of cell lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF Total SKP2 detection reagents were added. After an overnight incubation, the HTRF signal was recorded with an Envision Nexus reader.
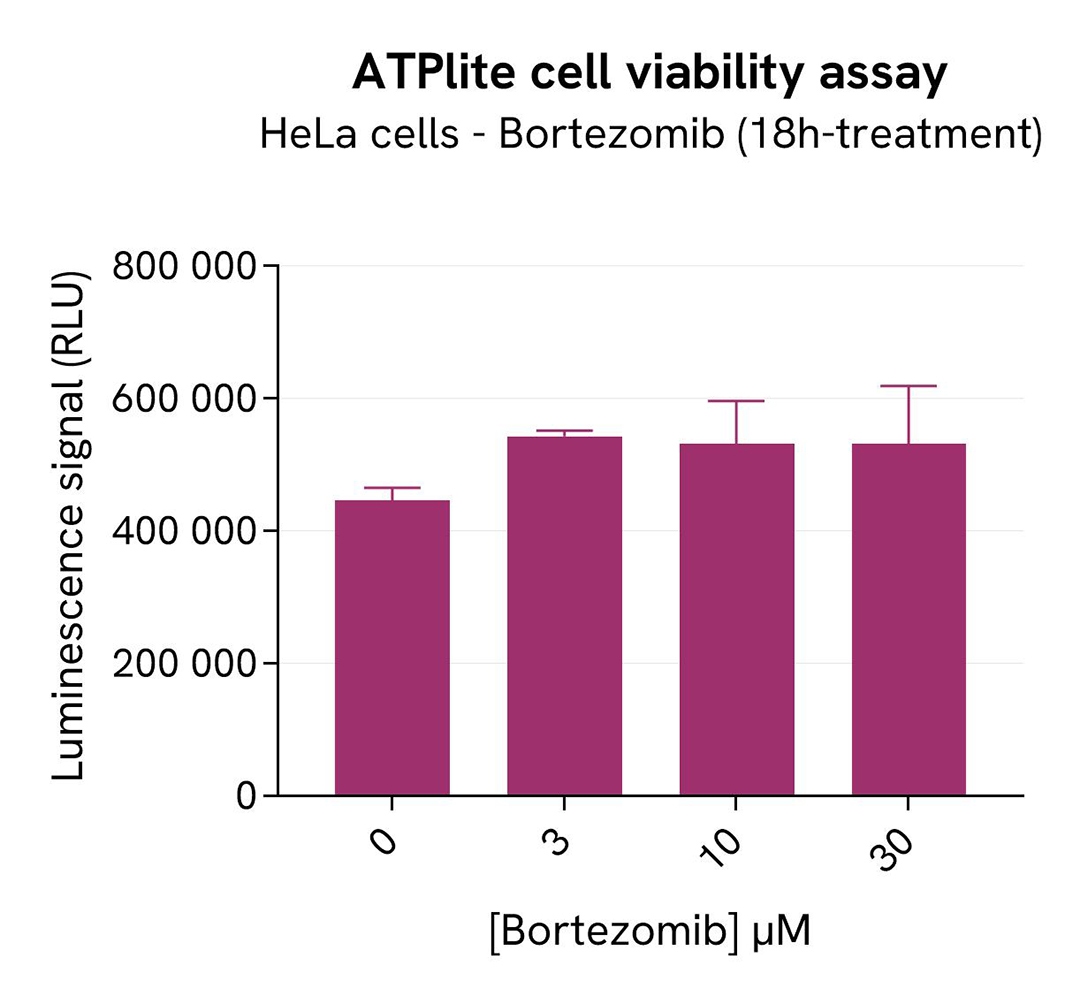
In parallel, cell viability was assessed with the ATPlite 1step Kit (Revvity®, #6016736/1/9). 5 µL of the same cell lysate were transferred into an HTRF 96-well low volume white plate, and 25 µL of ATPlite 1step detection reagent were added. After a 10-min incubation, the luminescence signal was measured with an Envision Nexus reader.
As expected, Bortezomib induced an SKP2 signal decrease. In addition, Bortezomib did not induce cytotoxic effects, as measured by the cell viability indicator ATPLite.
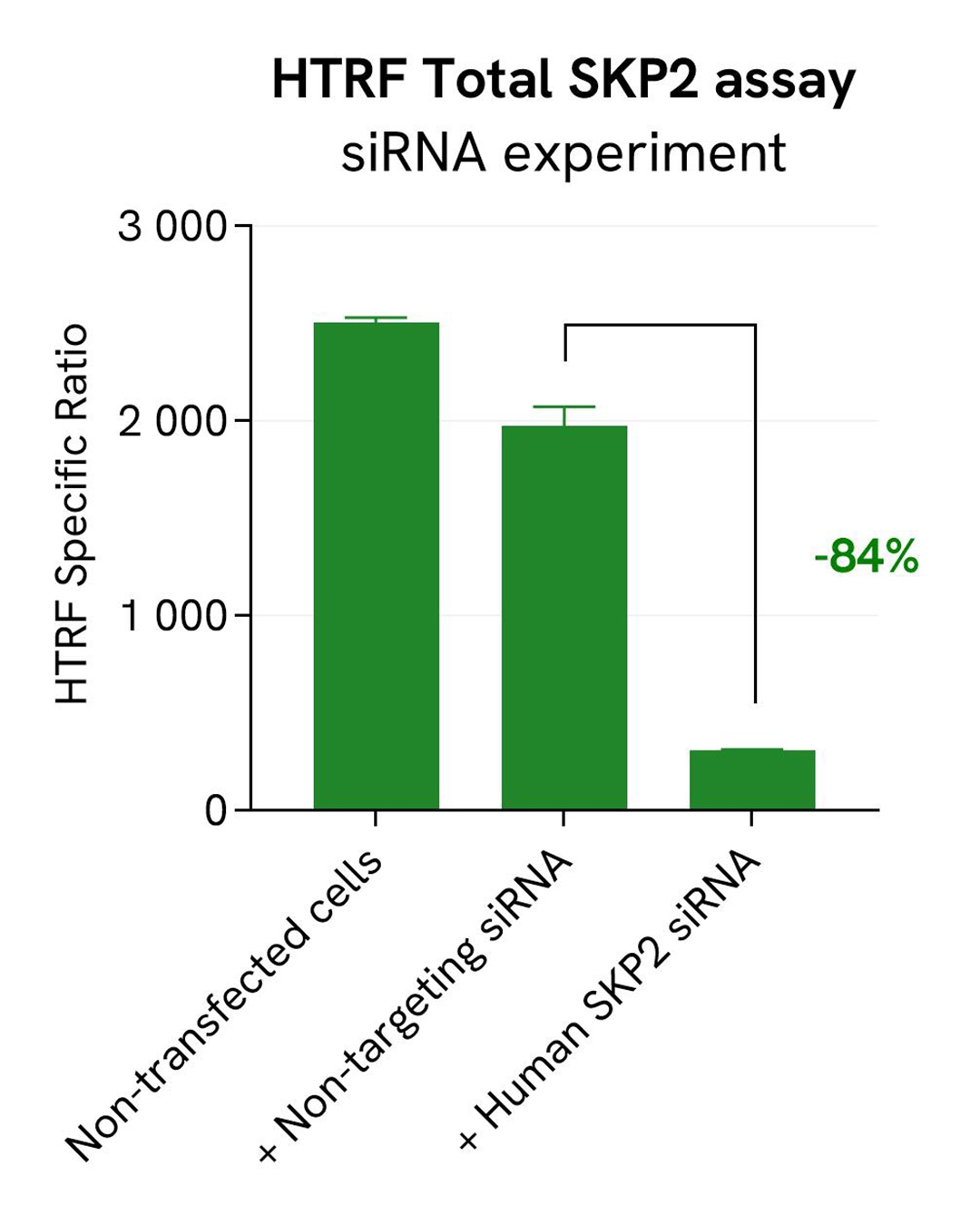
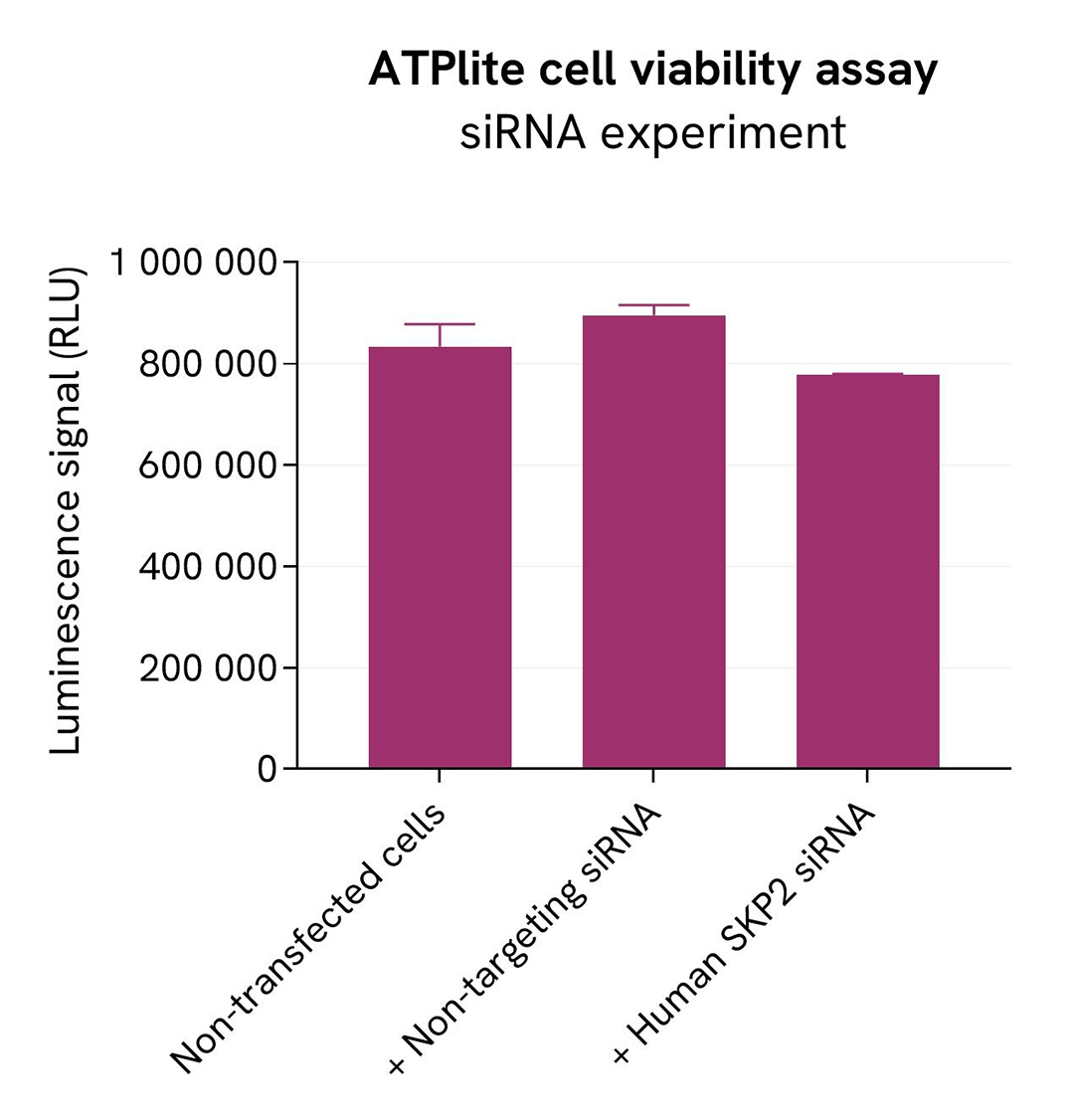
Specificity of Total SKP2 assay using siRNA
HeLa cells were seeded in a 96-well culture-treated plate (25,000 cells/well) in complete culture medium, and incubated overnight at 37°C, 5% CO2. The next day, the cells were transfected with ON-TARGETplus SMARTPool siRNA SKP2 (Horizon Discovery/Revvity) SKP2, as well as with a non-targeting siRNA used as negative control using DharmaFECT1 transfection reagent. After a 24h-incubation, the medium was replaced by fresh culture medium, and cells were incubated for an additional 24 hours. Following culture medium removal, cells were lysed with 25 µl of supplemented lysis buffer #1 (1X) + BR (1X) for 30 min at room temperature under gentle shaking.
Following cell lysis, 16 µL of cell lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF SKP2 detection antibodies were added. After an overnight incubation, the HTRF signal was recorded with an Envision Nexus reader.
In parallel, cell viability was assessed with the ATPlite 1step Kit (Revvity, #6016736/1/9). 5 µL of the same cell lysate were transferred into an HTRF 96-well low volume white plate, and 25 µL of ATPlite 1step detection reagent were added. After a 10-min incubation, the luminescence signal was measured with an Envision Nexus reader.
The siRNA experiment demonstrated that HTRF detection antibodies specifically measure SKP2. It was found that treatment with the SKP2 siRNA induced a dramatic HTRF signal loss. In addition, siRNA did not induce cytotoxic effects, as measured by the cell viability indicator ATPLite.
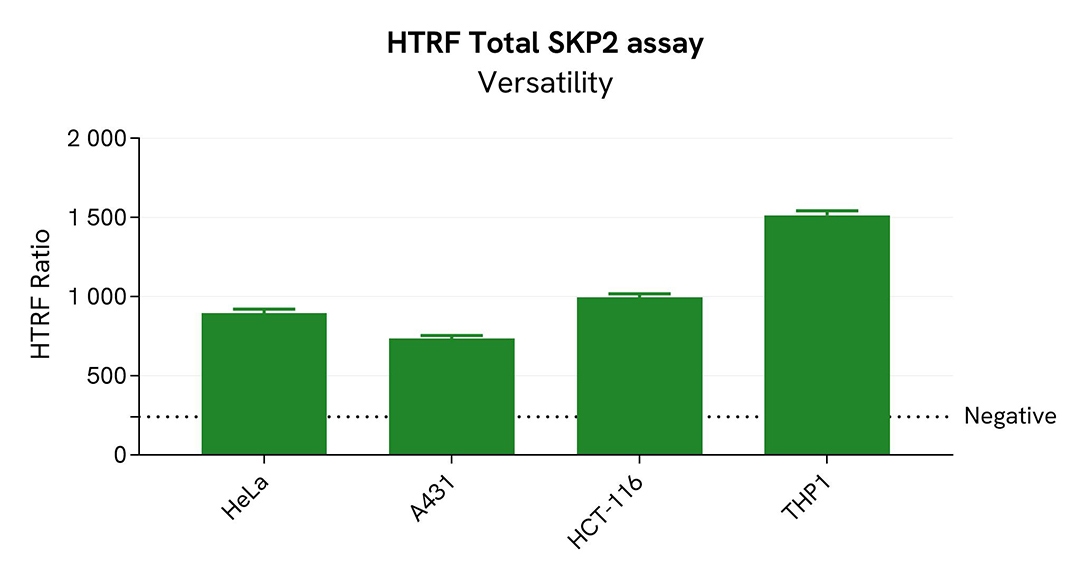
Assessment of total SKP2 level in various cell lines
HeLa, A431, HCT-116, and THP-1 cells were seeded in a 96-well culture-treated plate (25,000 cells/well for all cell lines except 20,000 cells/well for HCT-116) in complete culture medium, and incubated overnight at 37°C, 5% CO2. After culture medium removal, the cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at RT under gentle shaking.
For the detection step, 16 µL of cell lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF Total SKP2 detection reagents were added. After an overnight incubation, the HTRF signal was recorded with an Envision Nexus reader.
The HTRF Total SKP2 assay efficiently detected Total SKP2 in various cellular models expressing different levels of the protein.
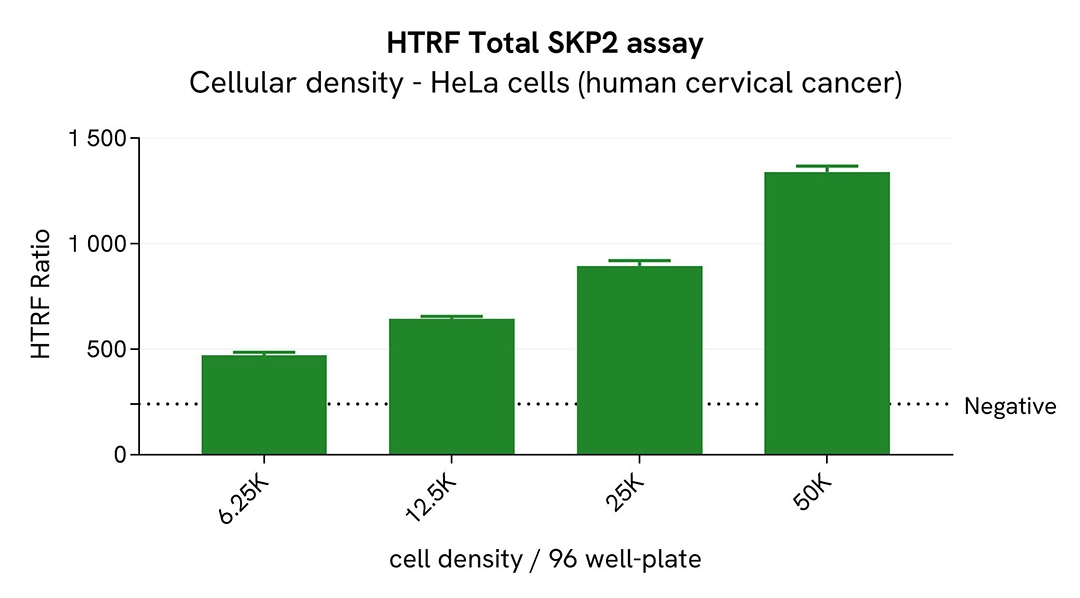
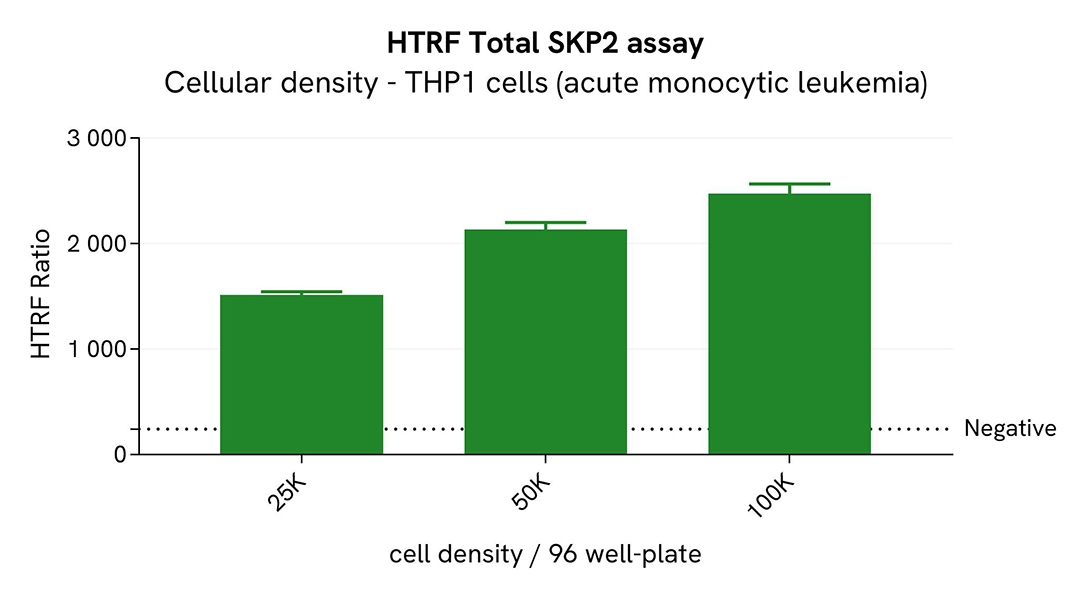
Broad dynamic range of Total SKP2 assay
Various cell densities of HeLa and THP-1 cells were seeded in a 96-well culture-treated plate in complete culture medium, and incubated overnight at 37°C, 5% CO2. After culture medium removal, the cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at RT under gentle shaking.
For the detection step, 16 µL of cell lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF Total SKP2 detection reagents were added. After an overnight incubation, the HTRF signal was recorded with an Envision Nexus reader.
The HTRF Total SKP2 assay has demonstrated a broad dynamic range.
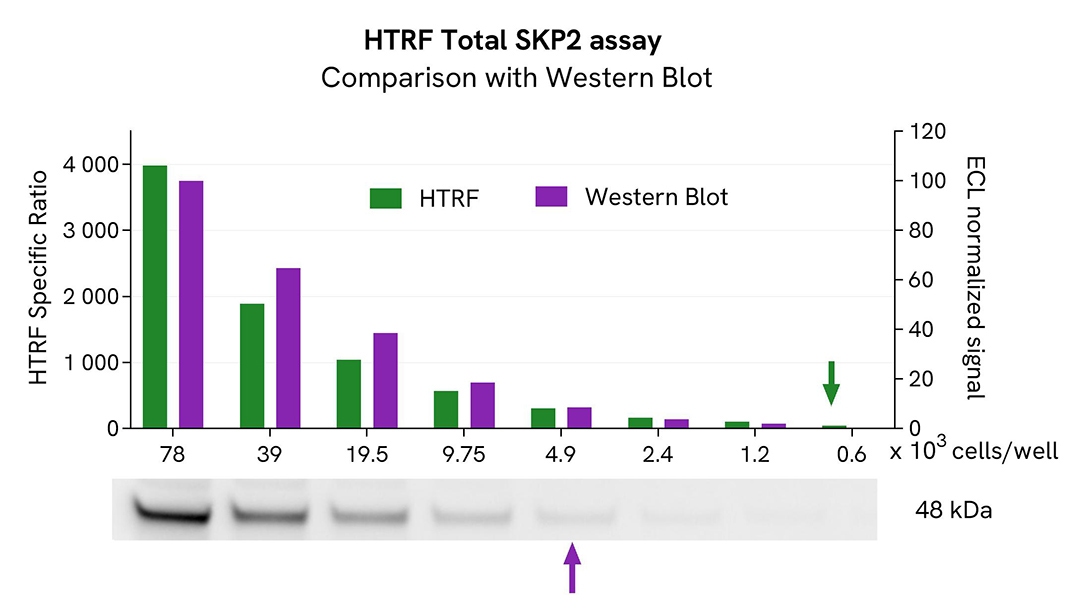
HTRF Total SKP2 assay compared to Western Blot
HeLa cells were cultured in flasks and incubated for 48h at 37°C, 5% CO2. After culture medium removal, cells were lysed with 3 mL of supplemented Lysis Buffer #1 for 30 min at room temperature under gentle shaking. Equal amounts of cell lysates (13 µL) were used for a side-by-side comparison of Western Blot and HTRF techniques.
Using the HTRF Total SKP2 assay, 600 cells/well were enough to detect a significant signal, while more than 4,900 cells were needed to obtain a minimal chemiluminescent signal using Western Blot. Therefore, in these conditions, the HTRF Total SKP2 assay is 8 times more sensitive than the Western Blot technique.
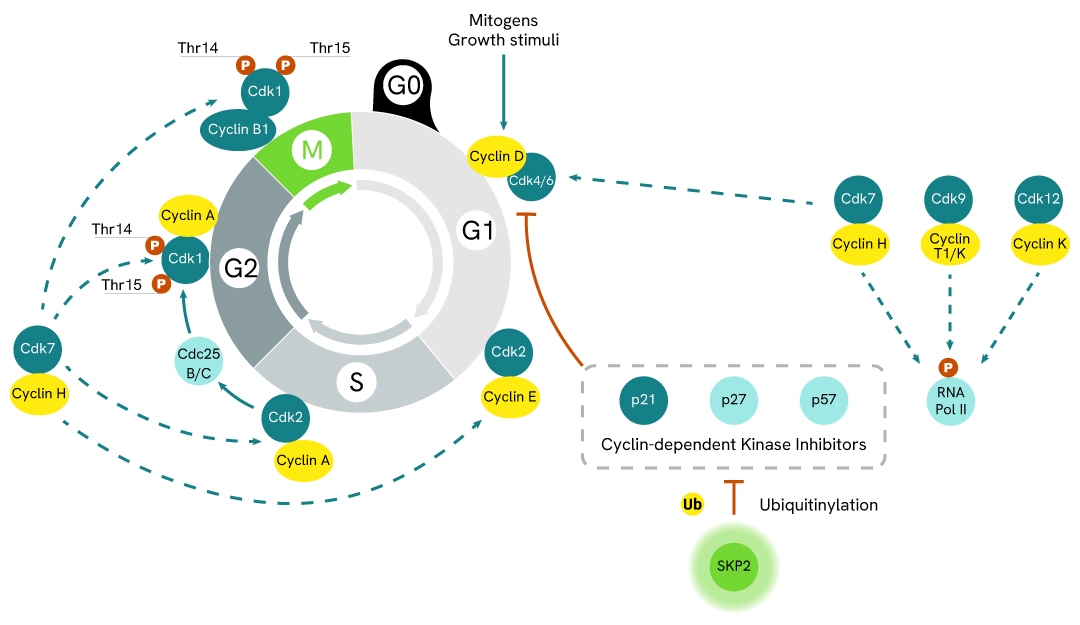
Simplified pathway
SKP2 signaling pathway
S-phase kinase-associated protein 2 (SKP2) is a protein with E3 ligase functions. It is involved in the regulation of the cell life cycle where it modulates the activity of Cyclin-dependent Kinase Inhibitors (CKIs) by promoting their ubiquitinylation and subsequent hydrolysis.
When ubiquitinylated by SKP2, inhibitors such as p21, p27, or p57 become less available, which promotes cyclin activity and the cell life cycle. Mitogenic signals, such as growth factors, trigger cells to enter the G1 phase of the cell cycle by inducing cyclin D synthesis. Cyclin D then interacts with CDK4 and CDK6 to form active complexes. Both activated kinases are then able to mono-phosphorylate the tumor suppressor RB (protein of retinoblastoma). This still binds to transcription factor E2F, but some genes can be transcribed, such as cyclin E. In the late G1 and early S phases, Cyclin E interacts with and activates CDK2. This in turn phosphorylates additional sites on RB, resulting in its complete inactivation. The E2F-responsive genes required for S phase progression are thus induced, such as Cyclin A, which then interacts with CDK2 to form Cyclin A/CDK2 complexes. CDK2 finally phosphorylates Cdc25B & Cdc25C phosphatases, which in turn activate CDK1, required for progression in the G2 and M phases of the cell-division cycle.
As such, SKP2 overactivation is a common driver of oncogenesis and drug resistance, due to its promotion of unregulated cell-division cycles.
Specifications
| Application |
Cell Signaling
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 1
Lysis Buffer 2
Lysis Buffer 4
|
| Molecular Modification |
Total
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target |
SKP2
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Immuno-oncology
Oncology
|
| Unit Size |
10,000 assay points
|
Resources
Are you looking for resources, click on the resource type to explore further.
Loading...


How can we help you?
We are here to answer your questions.






























