
HTRF Human and Mouse Total Heat Shock Protein 1A/1B (Hsp70 1A/1B) Detection Kit, 500 Assay Points
| Feature | Specification |
|---|---|
| Application | 細胞シグナル伝達 |
| Sample Volume | 16 µL |


Loading...
Product information
Overview
HSP70-1A and HSP70-1B, also known as HSPA1A and HSPA1B, are the major stress-inducible 70-kDa heat shock proteins. These highly conserved molecular chaperones play a critical role in maintaining protein quality control and cellular homeostasis under stressful conditions such as heat shock, oxidative stress, exposure to heavy metals, or proteotoxic drugs.
HSP70-1A/1B are also potent anti-apoptotic proteins, making them essential for the growth and survival of cancer cells. Elevated expression levels of these proteins are observed in various cancer types and are associated with poor prognosis, recurrence, and resistance to treatment. Consequently, targeting HSP70-1A/1B represents a promising strategy to sensitize tumors to therapy.
Conversely, HSP70-1A/1B have been shown to protect the brain from a range of nervous system injuries by inhibiting both cell death and immune responses. As a result, pharmaceutical compounds capable of inducing HSP70 expression are being explored as potential treatments for neurological diseases.
How it works
Total HSP70-1A/1B assay principle
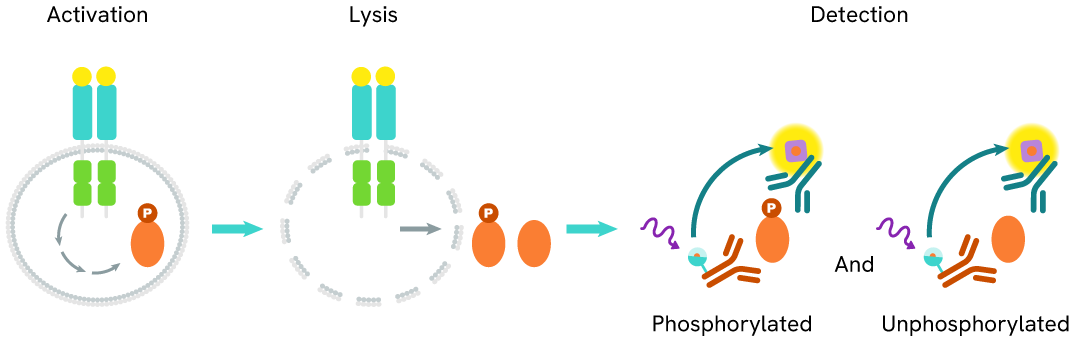
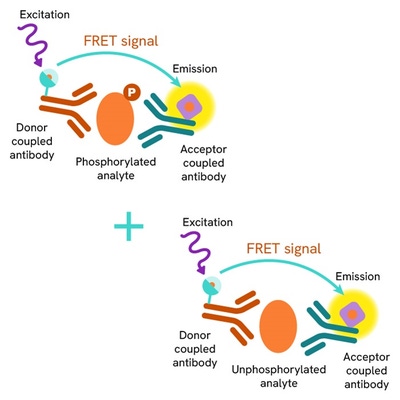
The Total HSP70-1A/1B assay measures the levels of HSP70-1A/1B in cells. Unlike Western Blot, this assay is entirely plate-based and does not require gels, electrophoresis, or protein transfer. It utilizes two antibodies: one labeled with a donor fluorophore and the other with an acceptor. Both antibodies are highly specific to distinct epitopes on the target protein.
In the presence of HSP70-1A/1B, an immune complex forms involving both labeled antibodies, bringing the donor fluorophore into close proximity with the acceptor. This interaction generates a FRET signal, the intensity of which is directly proportional to the concentration of total protein in the sample. The assay also enables assessment of the protein’s phosphorylation state, all within a no-wash assay format.

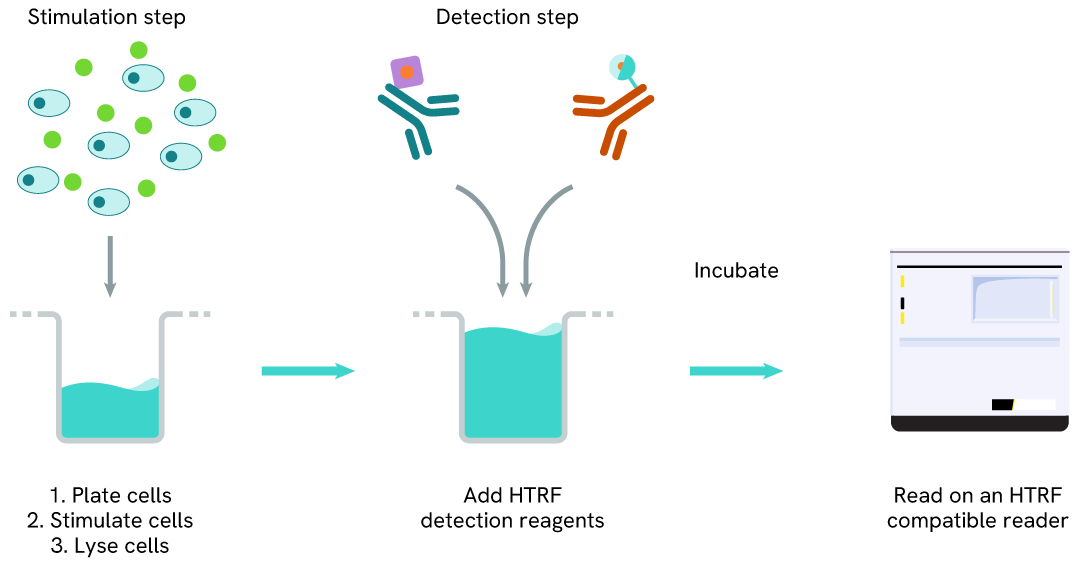
Total HSP70-1A/1B two-plate assay protocol
The two-plate protocol involves culturing cells in a 96-well plate before lysis, then transferring lysates into a 384-well low volume detection plate before the addition of Total HSP70-1A/1B HTRF detection reagents. This protocol allows for the cells' viability and confluence to be monitored.
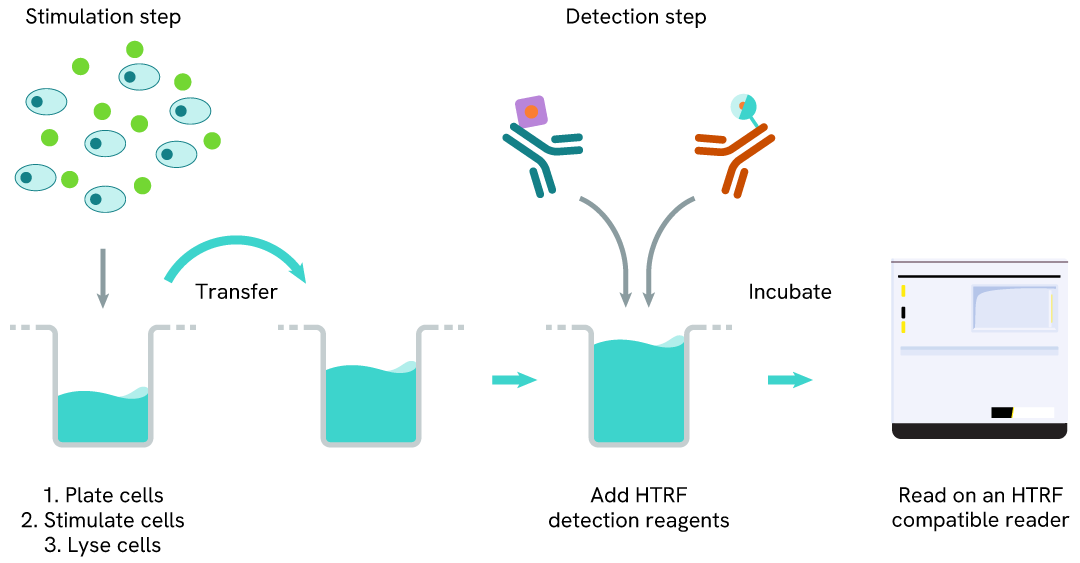
Total HSP70-1A/1B one-plate assay protocol
Detection of Total HSP70-1A/1B with HTRF reagents can be performed in a single plate used for culturing, stimulation, and lysis. No washing steps are required. This HTS designed protocol allows miniaturization while maintaining robust HTRF quality.
Assay validation
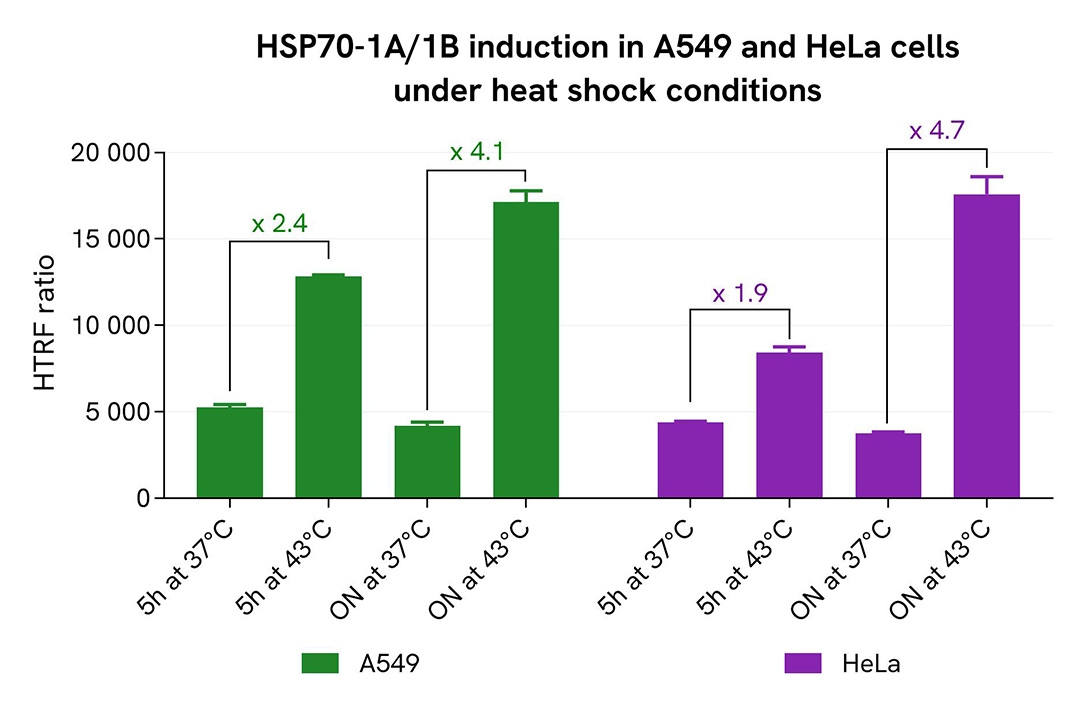
HSP70-1A/1B quantification following thermal stress
The human cancer cell lines A549 and HeLa were seeded in 96-well culture-treated plates (12,500 cells/well) in complete culture medium, and incubated for 5 hours at 37°C with 5% CO₂. The cells were then subjected to heat shock by incubation at either 37°C (control) or 43°C for 5 hours or overnight. Following the stress treatment, cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking.
For detection, 16 µL of cell lysate were transferred into a 384-well low-volume white microplate, and 4 µL of HTRF Total HSP70-1A/1B detection reagents were added. After a 3-hour incubation at room temperature, the HTRF signal was recorded using an EnVision Nexus reader.
Cell viability was assessed by transferring 5 µL of the same lysate into a 96-well low-volume white plate, followed by the addition of 25 µL of ATPlite 1step reagent (ATPlite 1step Luminescence Assay System, Cat #6016736/1/9). Luminescence was measured on the EnVision Nexus reader after a 10-minute incubation at room temperature in the dark.
As expected, both cell lines responded to heat shock with increased expression of HSP70-1A/1B. A 2-fold increase was observed after 5 hours at 43 °C, with induction reaching 4- to 5-fold following overnight thermal stress. These results were further supported by the ATPlite 1step assay, which showed no significant changes in cell viability (data not shown).
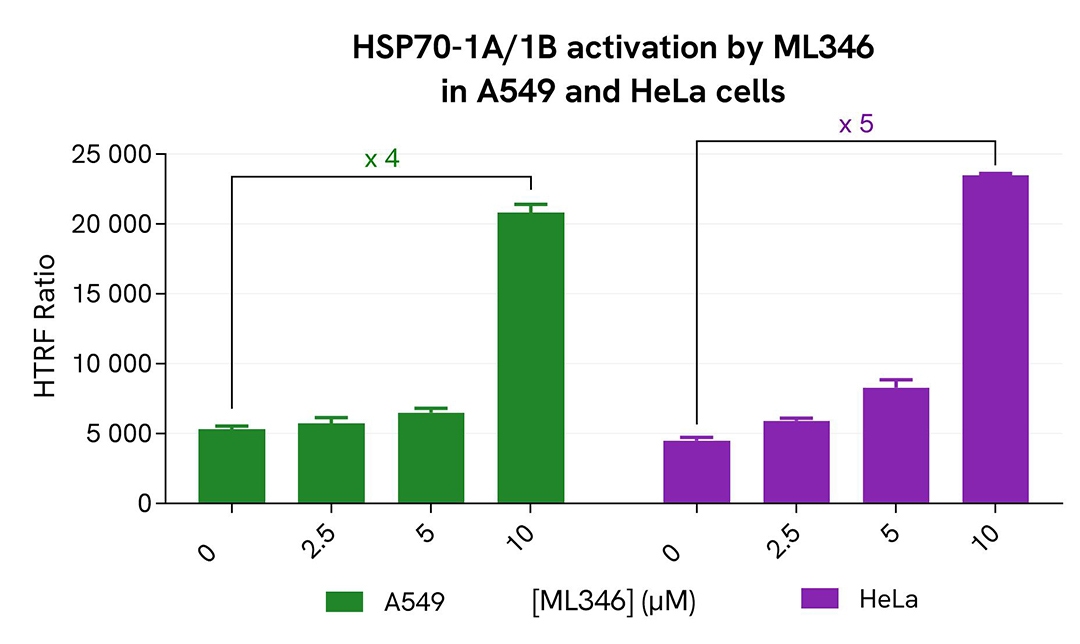
Quantification of HSP70-1A/1B expression following ML346 treatment
A549 and HeLa cells were seeded in 96-well culture-treated plates (12,500 cells/well) in complete culture medium, and incubated for 5 hours at 37°C with 5% CO₂. The cells were then treated overnight with increasing concentrations of the compound ML346. Following treatment, cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking.
For detection, 16 µL of lysate were used to quantify total HSP70-1A/1B levels using the HTRF kit, while 5 µL of the same lysate were used to assess cell viability using the ATPlite 1step Luminescence Assay System, as described in the previous section.
As expected, treatment with the HSP70 activator ML346 resulted in a dose-dependent induction of HSP70-1A/1B, with a 4-fold increase observed in A549 cells and a 5-fold increase in HeLa cells at the highest compound concentration. These findings were further supported by the ATPlite 1step assay, which showed no significant changes in cell viability (data not shown).
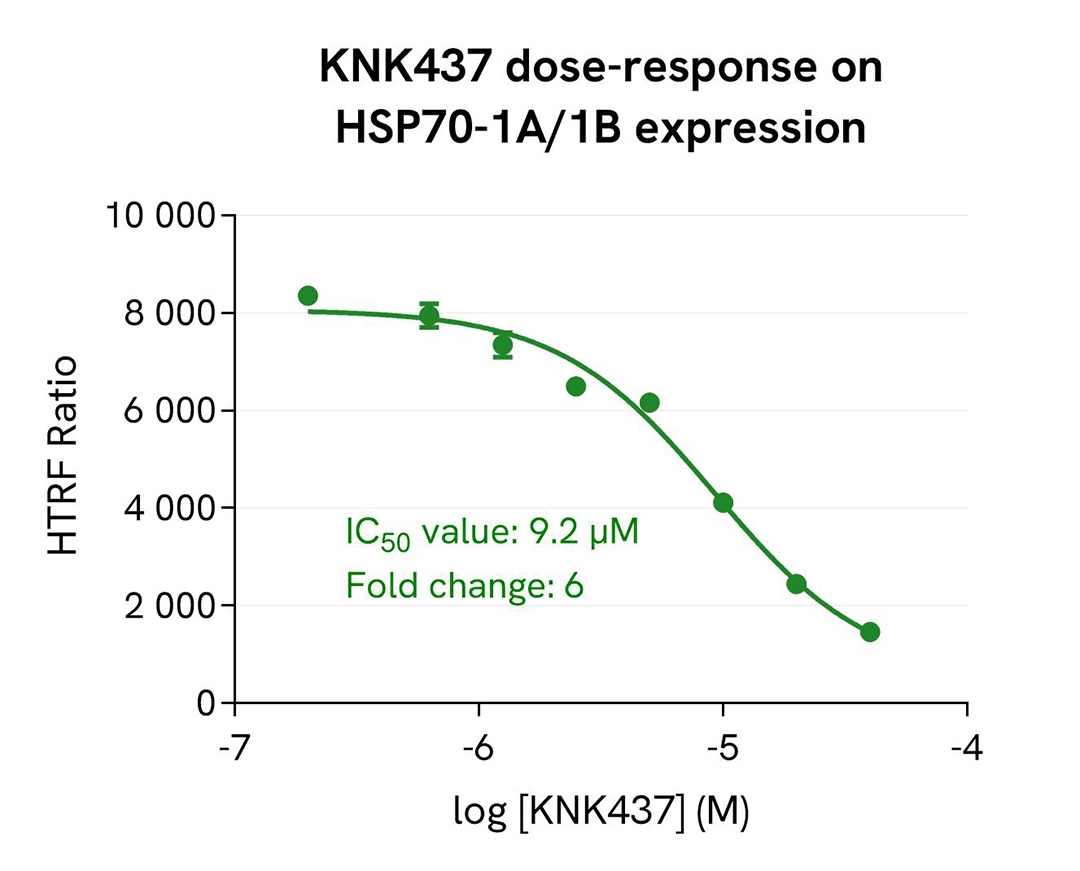
KNK437-mediated inhibition of HSP70-1A/1B induction in HeLa cells
HeLa cells were seeded in 96-well culture-treated plates (12,500 cells/well) in complete culture medium and incubated for 5 hours at 37°C with 5% CO₂. The cells were then pre-treated for 1 hour at 37°C with increasing concentrations of the compound KNK437, followed by an overnight heat shock at 43°C. After treatment, cells were lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking.
For detection, 16 µL of lysate were used to quantify total HSP70-1A/1B levels using the HTRF kit, and 5 µL of the same lysate were used to assess cell viability using the ATPlite 1step Luminescence Assay System, as described in the first section.
As expected, the heat shock protein inhibitor KNK437 induced a dose-dependent inhibition of heat-induced HSP70-1A/1B expression in HeLa cells, with an IC₅₀ value of 9.2 µM and a fold change of 6. These results were further supported by the ATPlite 1step assay, which showed no significant changes in cell viability (data not shown).
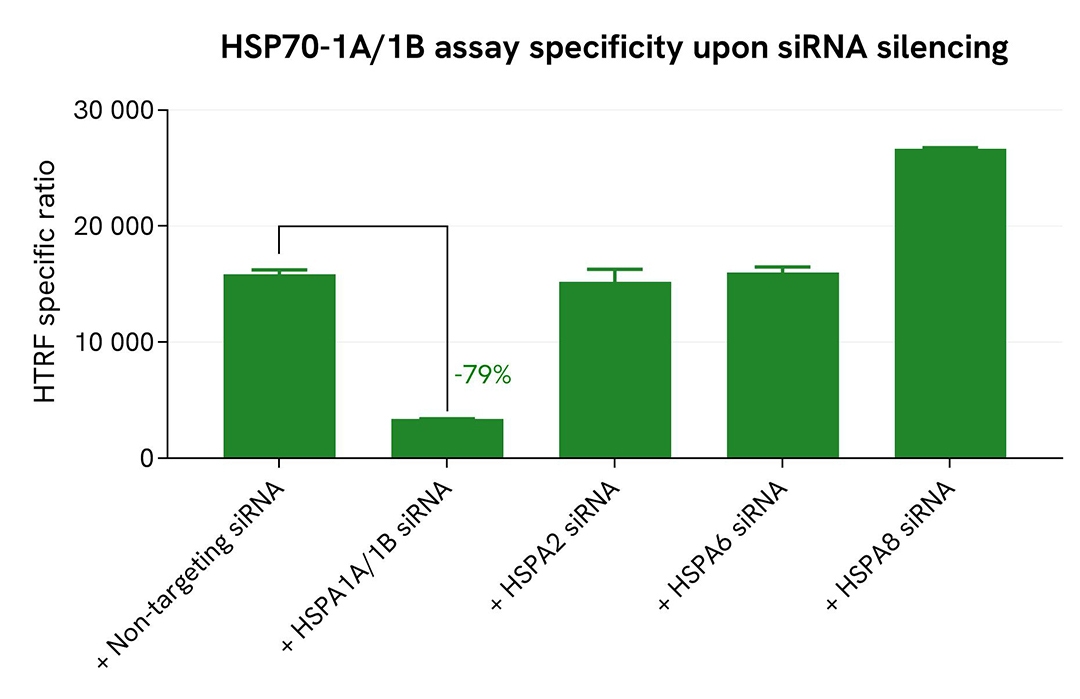
Validation of Total HSP70-1A/1B assay selectivity by siRNA experiments
A549 cells were seeded in 96-well culture-treated plates (25,000 cells/well in complete culture medium), and incubated overnight at 37°C with 5% CO₂. The following day, cells were transfected with ON-TARGETplus SMARTpool siRNAs (Horizon Discovery/Revvity) targeting human HSPA1A/1B (#L-005168-00-0010), HSPA2 (#L-010599-00-0010), HSPA6 (#L-019455-00-0010), or HSPA8 (#L-017609-00-0010), along with a non-targeting siRNA (#D-001810-10-20) used as a negative control. After 24 hours of siRNA incubation, the medium was replaced, and cells were incubated for an additional 24 hours. Cells were then lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking.
For detection, 16 µL of lysate were used to quantify total HSP70-1A/1B using the HTRF kit, and 5 µL of the same lysate were used to assess cell viability using the ATPlite 1step Luminescence Assay System, as described in the first section.
The siRNA experiments demonstrated that the HTRF detection antibodies specifically recognize HSP70-1A/1B and do not cross-react with the closely related family members HSPA2, HSPA6, or HSPA8. Silencing of HSPA1A/1B (targeting both 1A and 1B isoforms) resulted in a 79% decrease in signal, whereas knockdown of HSPA2 and HSPA6 had no effect on the assay signal. Interestingly, silencing of HSPA8 (encoding HSC70) led to a significant increase in signal, likely due to compensatory upregulation of HSP70-1A/1B, a well-documented cellular response (Koren et al., J. Biol. Chem. 2010, 285:2498–2505; Havik and Bramham, Oncol. Rep. 2007, 17:1501–1510). These findings were further supported by the ATPlite 1step assay, which showed no significant changes in cell viability in the presence of siRNAs (data not shown).
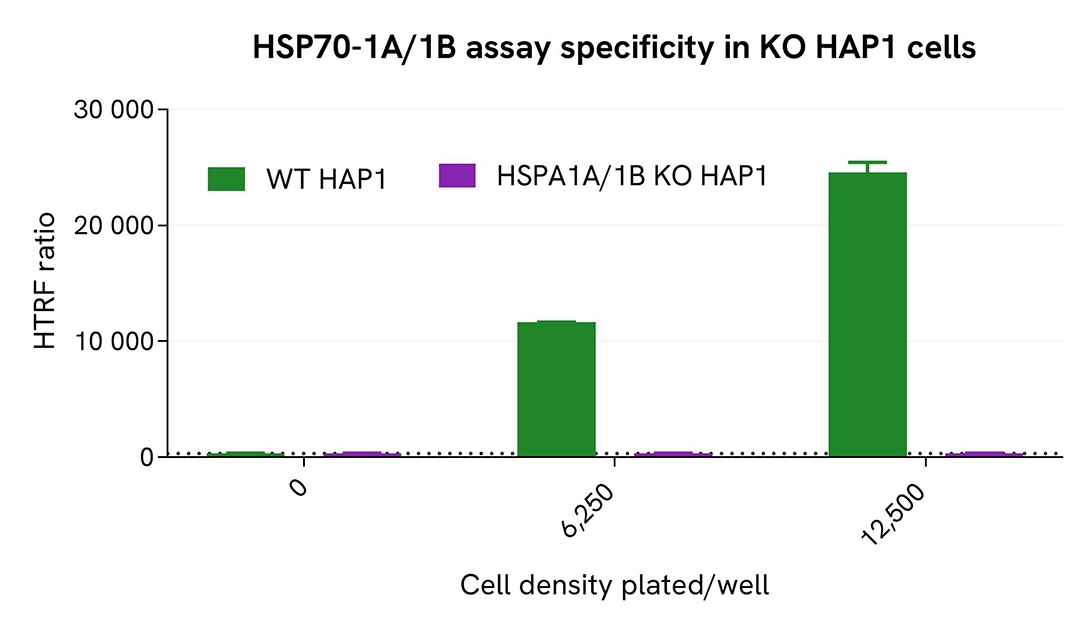
Validation of Total HSP70-1A/1B assay selectivity with HSPA1A/1B KO cells
The parental (wild-type, WT) HAP1 cell line and the HSPA1A/1B knockout (KO) HAP1 cell line (Horizon Discovery/Revvity, #HD HAP1-64507) were seeded in 96-well culture-treated plates (6,250 and 12,500 cells/well, respectively) in complete culture medium and incubated overnight at 37°C with 5% CO₂. Cells were then lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking.
For detection, 16 µL of lysate were used to quantify total HSP70-1A/1B using the HTRF kit, as described in the first section.
Total HSP70-1A/1B was clearly detected in WT HAP1 cells, while the HTRF signal was undetectable in HSPA1A/1B KO HAP1 cells, confirming the selectivity of the kit for detecting HSP70-1A/1B.
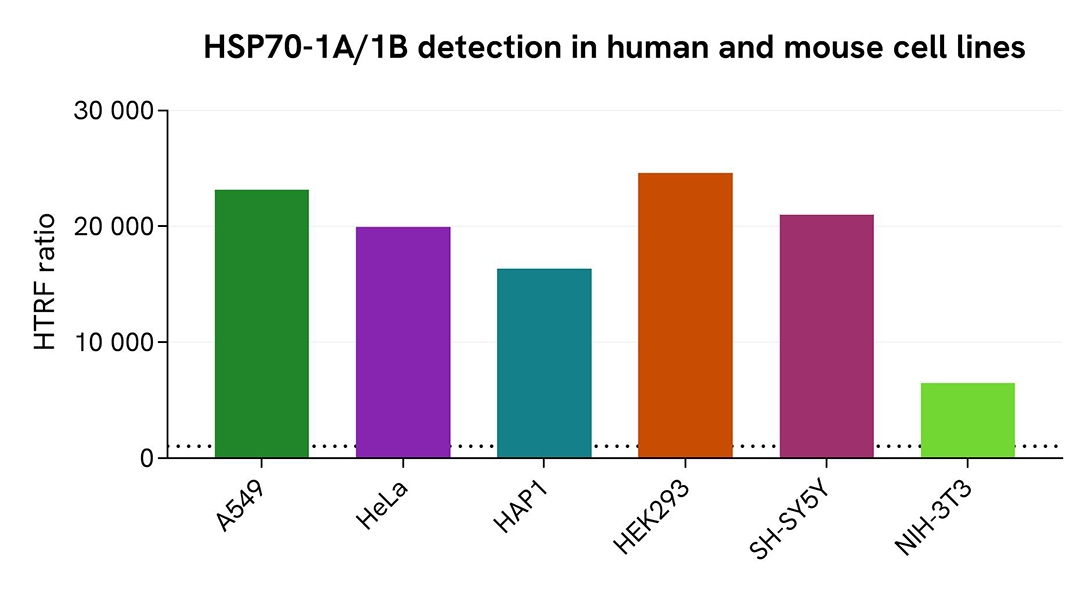
Validation of Total HSP70-1A/1B assay on different human and mouse cell lines
The human cell lines A549, HeLa, HAP1, HEK293, and SH-SY5Y, along with the mouse cell line NIH-3T3, were seeded in 96-well culture-treated plates (12,500 cells/well for the human cell lines and 100,000 cells/well for NIH-3T3) in complete culture medium and incubated overnight at 37°C with 5% CO₂. Cells were then lysed with 50 µL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking.
For detection, 16 µL of lysate were used to quantify total HSP70-1A/1B using the HTRF kit, as described in the first section.
The HTRF Total HSP70-1A/1B assay efficiently detected HSP70-1A/1B in various human cell lines, including cancer and neuronal models. The detection antibodies also successfully recognized HSP70-1A/1B in mouse cells.
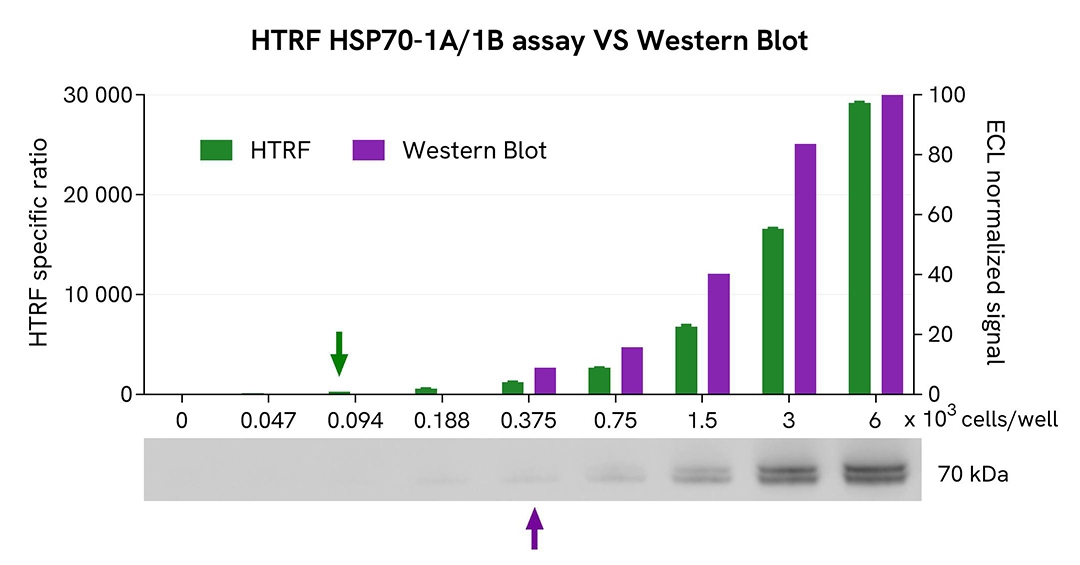
HTRF Total HSP70-1A/1B assay compared to Western Blot
HEK293 cells were cultured in a T175 flask in complete medium at 37°C with 5% CO₂ until approximately 80% confluency was reached. Cells were then lysed with 3 mL of supplemented lysis buffer #1 for 30 minutes at room temperature under gentle shaking.
Serial dilutions of the cell lysate were prepared using supplemented lysis buffer. For the HTRF assay, 16 µL of each dilution were transferred into a 384-well low-volume white microplate, followed by the addition of 4 µL of HTRF Total HSP70-1A/1B detection reagents. Equal amounts of lysate were also used for a side-by-side comparison with Western Blot.
Using the HTRF Total HSP70-1A/1B assay, a significant signal was detected with as few as 94 cells per well, whereas a minimum of 375 cells was required to obtain a detectable chemiluminescent signal by Western Blot. Under these conditions, the HTRF assay demonstrated a 4-fold higher sensitivity than Western Blot.
Simplified pathway
HSP70-1A/1B signaling pathway
HSP70-1A and HSP70-1B, encoded by the HSPA1A and HSPA1B genes, are the major stress-inducible 70-kDa heat shock proteins in humans. These highly conserved molecular chaperones are essential cytoprotective proteins that maintain protein quality control and cellular homeostasis in response to extreme physiological, pathological, and environmental stressors.
Under stress conditions such as heat shock, oxidative stress, exposure to heavy metals, or proteotoxic drugs, the transcription factor HSF1 is activated and converted into its DNA-binding form. It then translocates to the nucleus, where it induces transcription from the HSPA1A/1B promoter, leading to a rapid upregulation of HSP70-1A/1B expression.
As major orchestrators of the heat shock response (HSR), HSP70-1A/1B play a central role in proteostasis by maintaining the functional integrity of intracellular proteins. They promote protein stability by facilitating the proper folding and refolding of denatured or damaged proteins and by preventing protein aggregation. Alternatively, they can target misfolded proteins for degradation via the ubiquitin-proteasome system (UPS) or autophagy.
Beyond their role in protein homeostasis, HSP70-1A/1B also exert cytoprotective effects by regulating apoptosis and cell proliferation. Their upregulation has been shown to inhibit apoptotic and necroptotic pathways and to stabilize inhibitors of apoptosis proteins (IAPs). As a result, HSP70-1A/1B are critical for the survival and proliferation of cancer cells.

Specifications
| Application |
Cell Signaling
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 1
|
| Molecular Modification |
Total
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target |
HSP70 1A/1B
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
Mouse
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Central Nervous System
Oncology
|
| Unit Size |
500 assay points
|
Resources
Are you looking for resources, click on the resource type to explore further.
Loading...


How can we help you?
We are here to answer your questions.






























