
AlphaLISA Human and Mouse α-SMA Detection Kit, 5,000 Assay Points
The AlphaLISA™ α-SMA detection kit is designed for the simple and rapid quantification of α-Smooth Muscle Actin produced by cells, providing a fast no-wash alternative to traditional wash-based ELISA assays.
| Feature | Specification |
|---|---|
| Application | タンパク質定量 |
| Protocol Time | 3h at RT |
| Sample Volume | 5 µL |
The AlphaLISA™ α-SMA detection kit is designed for the simple and rapid quantification of α-Smooth Muscle Actin produced by cells, providing a fast no-wash alternative to traditional wash-based ELISA assays.


Loading...
Product information
Overview
Alpha-Smooth Muscle Actin or α-SMA is a member of Actin family that forms microfilaments and is used as a marker of myofibroblast formation. After tissue injury, TGF-beta locally released by inflammatory cells activates resident fibroblasts or quiescent HSCs (hepatic stellate cells). This leads to their differentiation into myofibroblasts, whose role is to migrate into the damaged tissue and synthesize ECM (extracellular matrix) components to repair the wound. Myofibroblasts are characterized by de novo expression of alpha-SMA, which is incorporated into actin stress fibers and confers a high contractile activity to the cells. Chronic tissue injury leads to persistent de novo formation of myofibroblasts (alpha-SMA+), excessive contraction, and deposition of ECM, eventually leading to tissue fibrosis.
Formats:
- Our 100 assay point kit allows you to run 100 wells in 96-well format, using a 100 µL reaction volume (10 µL of sample).
- Our 500 assay point kit allows you to run 500 wells in 96-well or 384-well format, using a 50 µL reaction volume (5 µL of sample).
- Our 5,000 assay point kit allows you to run 5,000 wells in 96-well or 384-well format, using a 50 µL reaction volume (5 µL of sample).
Features:
- No-wash steps, no separation steps
- ELISA alternative technology
- Sensitive detection
- Broad sample compatibility
- Small sample volume
- Results in less than 4 hours
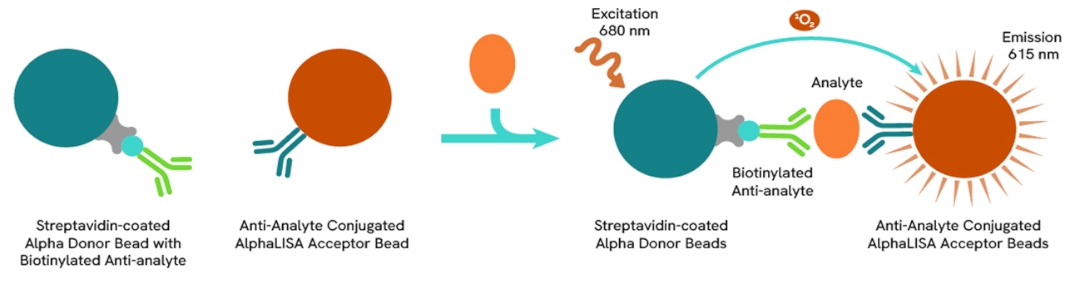
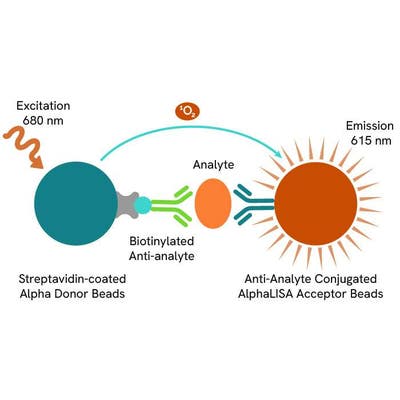
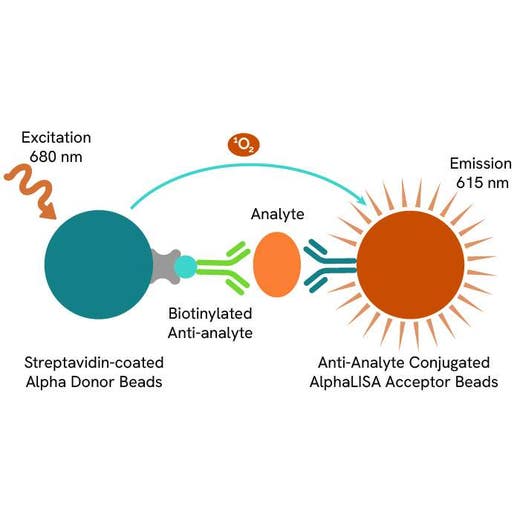
AlphaLISA technology allows the detection of molecules of interest in a no-wash, highly sensitive, quantitative assay. In an AlphaLISA assay, a biotinylated anti-analyte antibody binds to the Streptavidin-coated Donor beads while another anti-analyte antibody is conjugated to AlphaLISA Acceptor beads. In the presence of the analyte, the beads come into close proximity. The excitation of the Donor beads causes the release of singlet oxygen molecules that triggers a cascade of energy transfer in the Acceptor beads, resulting in a sharp peak of light emission at 615 nm.
How it works
Principle of the AlphaLISA human & mouse α-SMA assay
The AlphaLISA α-SMA assay is based on an AlphaLISA sandwich immunoassay involving a biotinylated anti-target antibody bound to Streptavidin-coated AlphaLISA Donor beads and an anti-target antibody conjugated to AlphaLISA Acceptor beads. Two antibodies are directed against the α-SMA protein. In the presence of the target, both antibodies bind to α-SMA and the beads come into proximity. The excitation of the Donor beads provokes the release of singlet oxygen molecules that triggers a cascade of energy transfer within the Acceptor beads, resulting in emission with λmax at 615 nm. The intensity of the signal is directly proportional to the concentration of α-SMA present in the sample (cell lysate).

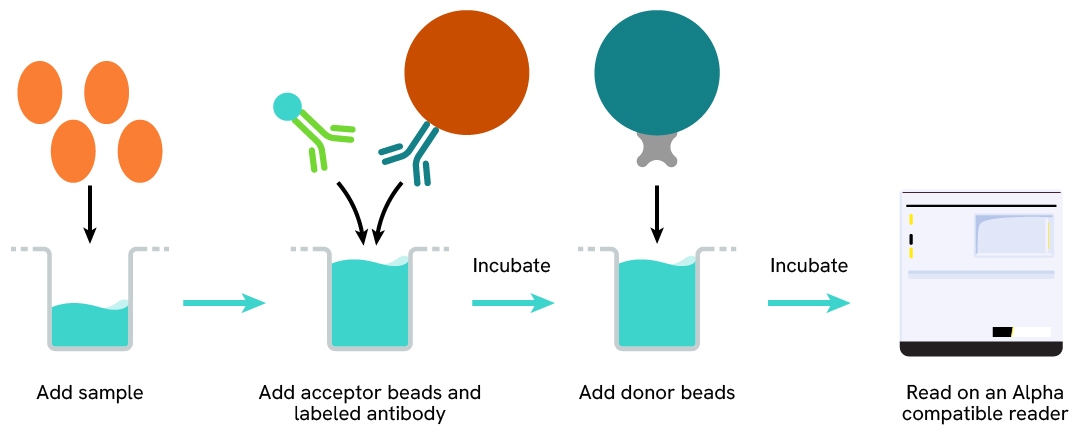
Protocol of the AlphaLISA human & mouse α-SMA assay
The AlphaLISA α-SMA assay can be run in a 96- or 384-well detection plate (50 µL final). As described here, samples (cell lysates) or control lysate are dispensed directly into the assay plate for the detection of α-SMA by AlphaLISA reagents. No washing steps are needed. The protocol can be further miniaturized or upscaled by simply resizing each addition volume proportionally.
Assay details
Human & mouse α-SMA assay details
| Kit components | Lyophilized analyte, SA-Donor beads, Biotinylated anti-analyte, Anti-analyte conjugated acceptor beads, Assay buffer 1 |
|---|---|
| Species | Human & mouse |
Assay validation
Selectivity for α-SMA isoform validated by siRNA experiments
The mouse fibroblast cell line NIH-3T3 was plated in culture-treated 96-well plates (10,000 cells/well) and incubated for 24h at 37°C - 5% CO2. The day after, the cells were transfected with an α-SMA siRNA or with a negative control siRNA for 24h and then treated or not with 5 ng/mL of TGF-ß1 for an additional 24 hours. After medium removal, the cells were lysed with 50 µL of lysis buffer for 30 minutes at RT under gentle shaking.
5 µL of lysate were transferred into a 384-well microplate before the sequential addition of AlphaLISA α-SMA detection reagents. Transfection with the α-SMA siRNA led to a huge decrease of the AlphaLISA specific signal (~ 90%) compared to the cells transfected with the negative control, demonstrating that the AlphaLISA α-SMA assay is specific for α-SMA.

MRC-5 differentiation into myofibroblasts in lung fibrosis
Human lung fibroblasts MRC-5 cells were plated in a culture-treated 96-well plate (25,000 cells/well) in complete culture medium. Once adhered, the supernatant was discarded and the cells were stimulated with increasing concentrations of TGF-ß in serum-free culture medium supplemented with 0.1% BSA for 24h at 37°C - 5% CO2.
After medium removal, cells were lysed with 50 µL of lysis buffer 1X for 30 minutes at RT under gentle shaking, and 5 µL of lysate were transferred into a 384-well plate before adding AlphaLISA α-SMA detection reagents. The treatment of cells with TGF-ß results in an 8.5-fold increase in α-SMA expression level, highlighting the differentiation of MRC-5 into myofibroblasts.

α-SMA expression upon differentiation of fibroblasts into myofibroblasts
The mouse fibroblast cell line NIH-3T3 was plated in a culture-treated 96-well plate (20,000 cells/well) in complete culture medium and incubated at 37°C - 5% CO2. The day after, the cells were treated with increasing concentrations of TGF-ß for 48 hours in serum-free culture medium supplemented with 0.1% BSA. After medium removal, the cells were lysed with 200 µL of lysis buffer 1X for 30 minutes at RT under gentle shaking, and 5 µL of lysate were transferred into a 384-well plate before the addition of AlphaLISA α-SMA detection reagents.
TGF-ß long-term treatment results in a three-fold increase of α-SMA expression level, which demonstrates the differentiation of fibroblasts into myofibroblasts.

Detection of α-SMA expression into cancer cell line
The Human cancer cell line HepG2 was plated in a culture-treated 96-well plate (from 10,000 to 40,000 cells/well) in complete culture medium and incubated at 37°C - 5% CO2 for 24h.
The day after, the supernatant was removed, and the cells were lysed with 50 µL of lysis buffer 1X for 30 minutes at RT under gentle shaking. For detection, 5 µL of lysate were transferred into a 384-well plate before the addition of AlphaLISA α-SMA detection reagents. As expected, α-SMA is well detected proportionally to the cellular densities in this oncological model.

Specifications
| Application |
Protein Quantification
|
|---|---|
| Automation Compatible |
Yes
|
| Brand |
AlphaLISA
|
| Detection Modality |
Alpha
|
| Product Group |
Kit
|
| Protocol Time |
3h at RT
|
| Sample Volume |
5 µL
|
| Shipping Conditions |
Shipped in Blue Ice
|
| Target |
α-SMA
|
| Target Class |
Biomarkers
|
| Target Species |
Human
Mouse
|
| Technology |
Alpha
|
| Therapeutic Area |
Cardiovascular
NASH/Fibrosis
|
| Unit Size |
5,000 assay points
|
Resources
Are you looking for resources, click on the resource type to explore further.
Alpha technolgy enables the rapid and straightforward mesaure of virtually any target. This includes enzymes, receptor-ligand...
This document includes detailed tables listing HTRF™, AlphaLISA™ SureFire® Ultra™, and Alpha SureFire® Ultra™ Multiplex assays...
Loading...


How can we help you?
We are here to answer your questions.






























