
AlphaLISA SureFire Ultra Human Phospho-IRF7 (Ser477) Detection Kit, 50,000 Assay Points
The AlphaLISA™ SureFire® Ultra™ Human Phospho-IRF7 (Ser477) assay is a sandwich immunoassay for quantitative detection of phospho-IRF7 (Ser477) in cellular lysates using Alpha Technology.
| Feature | Specification |
|---|---|
| Application | 細胞シグナル伝達 |
| Protocol Time | 2h at RT |
| Sample Volume | 10 µL |
The AlphaLISA™ SureFire® Ultra™ Human Phospho-IRF7 (Ser477) assay is a sandwich immunoassay for quantitative detection of phospho-IRF7 (Ser477) in cellular lysates using Alpha Technology.



Loading...
Product information
Overview
Interferon Regulatory Factor 7 (IRF7) is a master regulator of type I interferon (IFN) responses, particularly during viral infections. Upon activation by pattern recognition receptors (PRRs) such as TLR7, TLR9, and RIG-I, IRF7 undergoes phosphorylation, dimerization, and nuclear translocation, where it drives the transcription of IFN-α and IFN-β genes. IRF7 plays a central role in antiviral immunity, bridging innate immune sensing with the activation of adaptive immune responses. Beyond infection, dysregulated IRF7 activity is involved in autoimmunity, including systemic lupus erythematosus (SLE), and contributes to the immunopathology of certain cancers. Modulating IRF7 activity holds promise in antiviral therapies, cancer immunotherapy, and autoimmunity management.
The AlphaLISA SureFire Ultra Human Phospho-IRF7 (Ser477) Detection Kit is a sandwich immunoassay for the quantitative detection of phospho-IRF7 (Ser477) in cellular lysates, using Alpha Technology.
Formats:
- The HV (high volume) kit contains reagents to run 100 wells in 96-well format, using a 60 μL reaction volume.
- The 500-point kit contains enough reagents to run 500 wells in 384-well format, using a 20 μL reaction volume.
- The 10,000-point kit contains enough reagents to run 10,000 wells in 384-well format, using a 20 μL reaction volume.
- The 50,000-point kit contains enough reagents to run 50,000 wells in 384-well format, using a 20 μL reaction volume.
AlphaLISA SureFire Ultra kits are compatible with:
- Cell and tissue lysates
- Antibody modulators
- Biotherapeutic antibodies
AlphaLISA SureFire Ultra kits can be used for:
- Cellular kinase assays
- Receptor activation studies
- High-throughput screening for preclinical studies
How it works
Phospho-AlphaLISA SureFire Ultra assay principle
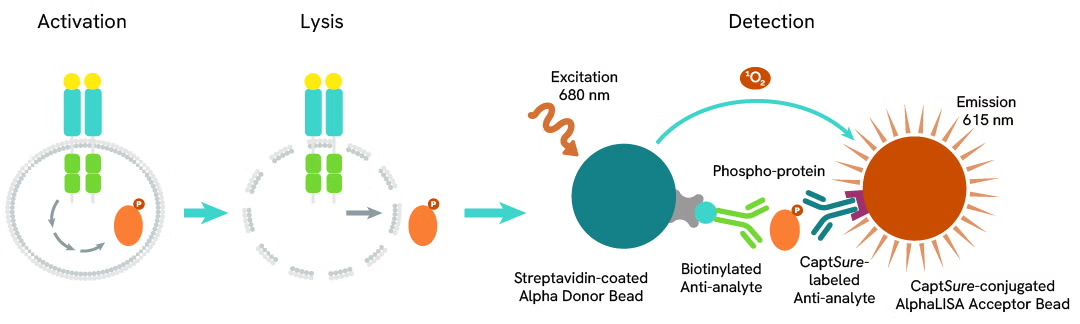
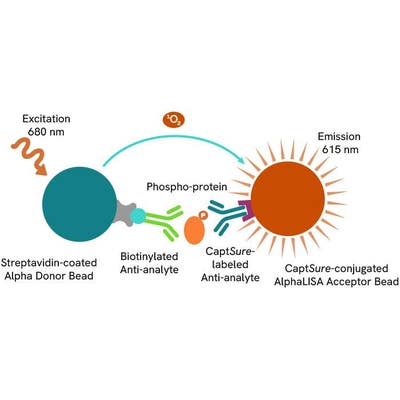
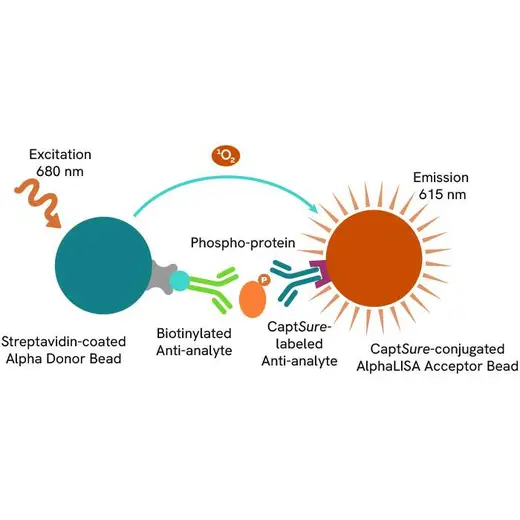
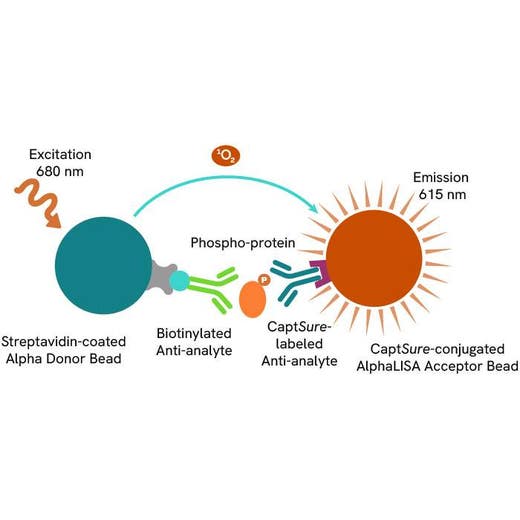
The Phospho-AlphaLISA SureFire Ultra assay measures a protein target when phosphorylated at a specific residue.
The assay uses two antibodies which recognize the phospho epitope and a distal epitope on the targeted protein. AlphaLISA assays require two bead types: Acceptor and Donor beads. Acceptor beads are coated with a proprietary CaptSure™ agent to specifically immobilize the assay specific antibody, labeled with a CaptSure tag. Donor beads are coated with streptavidin to capture one of the detection antibodies, which is biotinylated. In the presence of phosphorylated protein, the two antibodies bring the Donor and Acceptor beads in close proximity whereby the singlet oxygen transfers energy to excite the Acceptor bead, allowing the generation of a luminescent Alpha signal. The amount of light emission is directly proportional to the quantity of phosphoprotein present in the sample.

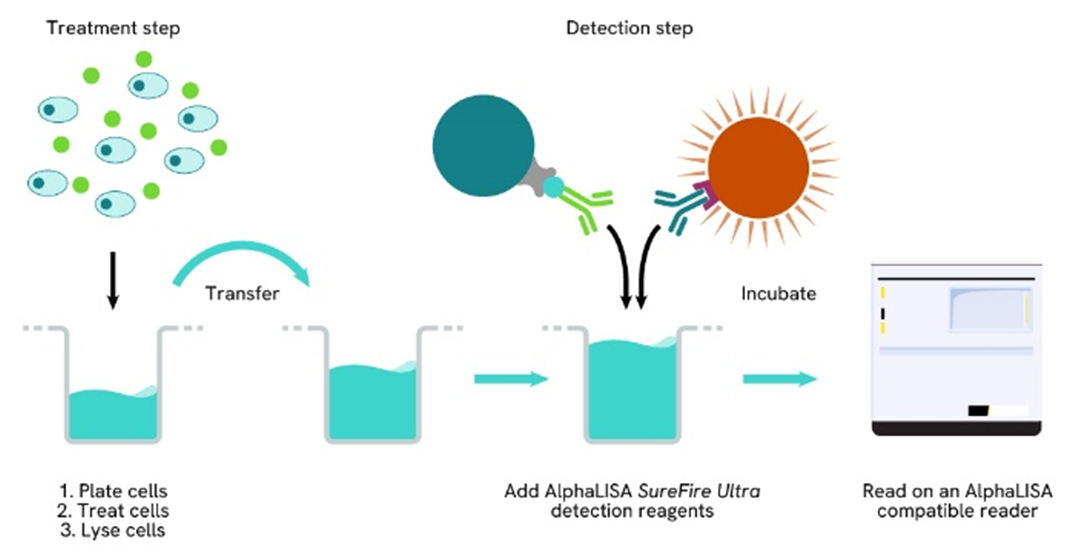
Phospho-AlphaLISA SureFire Ultra two-plate assay protocol
The two-plate protocol involves culturing and treating the cells in a 96-well plate before lysis, then transferring lysates into a 384-well OptiPlate™ plate before the addition of Phospho-AlphaLISA SureFire Ultra detection reagents. This protocol permits the cells viability and confluence to be monitored. In addition, lysates from a single well can be used to measure multiple targets.
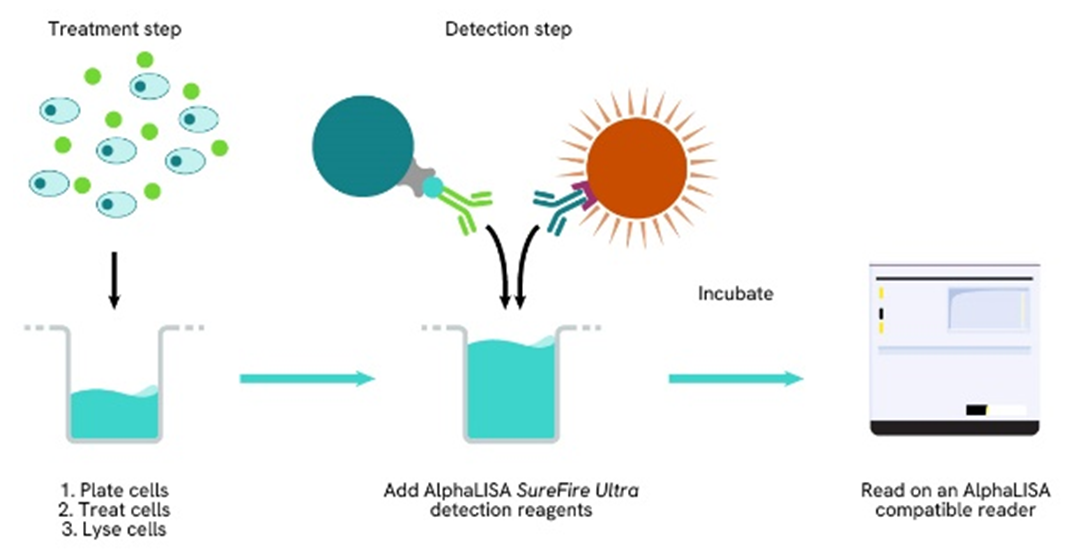
Phospho-AlphaLISA SureFire Ultra one-plate assay protocol
Detection of Phosphorylated target protein with AlphaLISA SureFire Ultra reagents can be performed in a single plate used for culturing, treatment, and lysis. No washing steps are required. This HTS designed protocol allows for miniaturization while maintaining AlphaLISA SureFire Ultra quality.
Assay validation
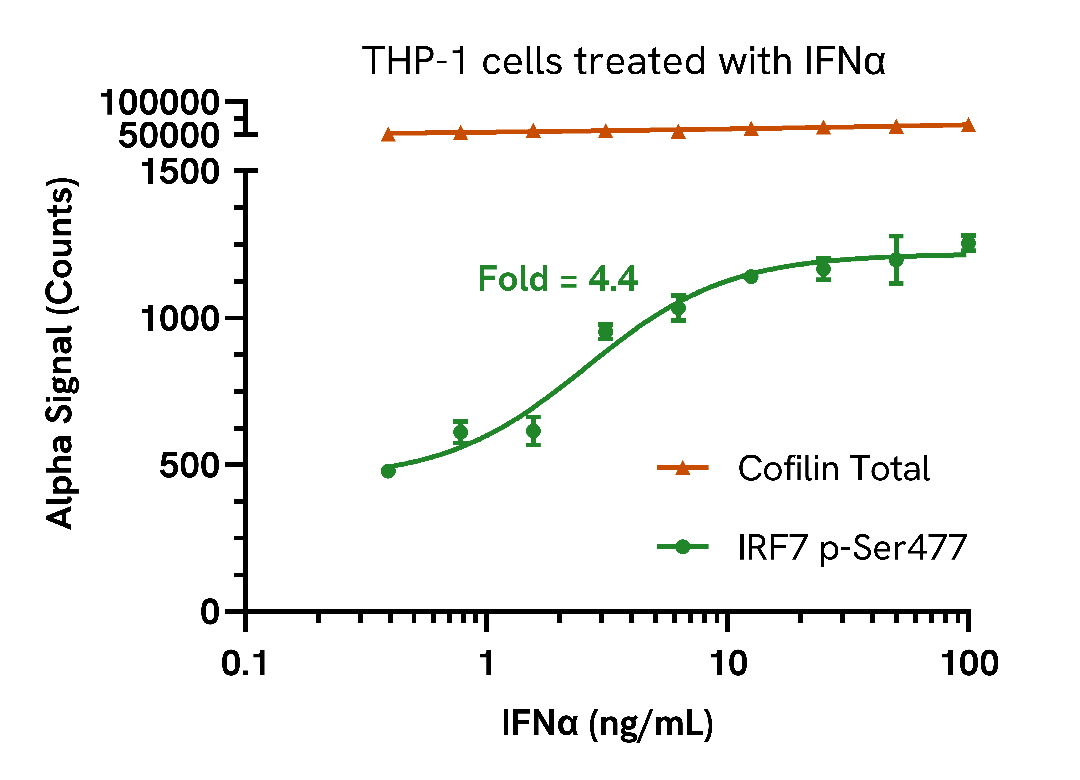
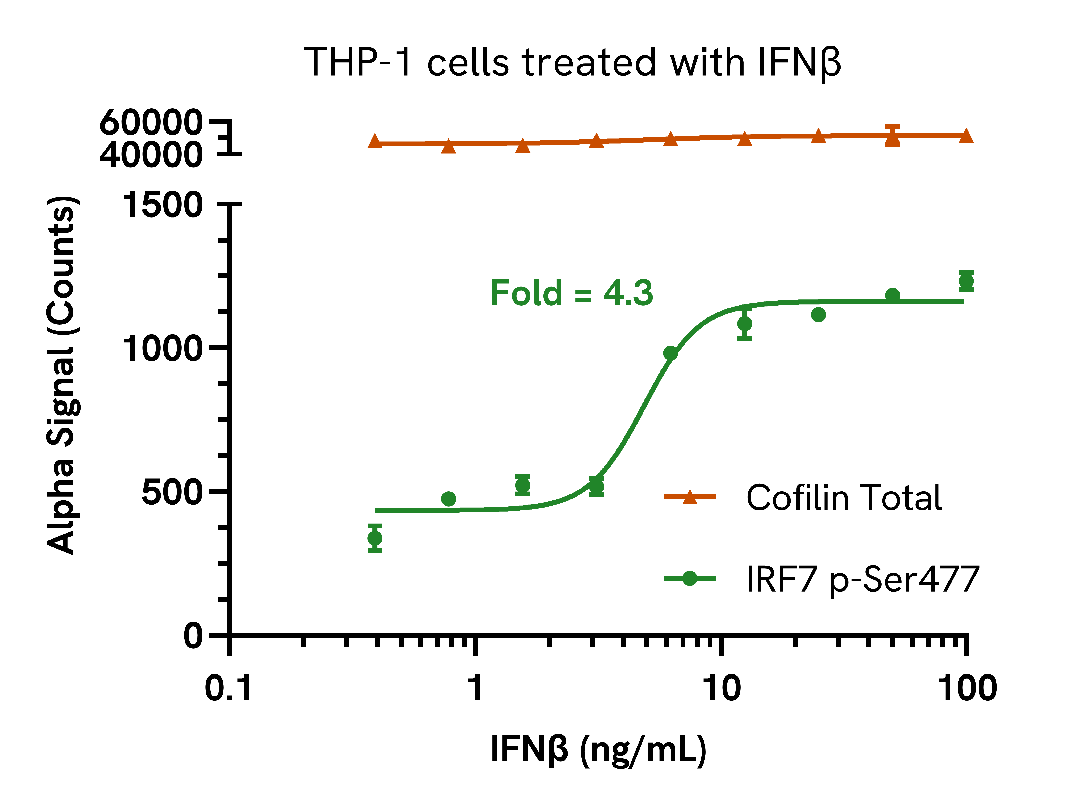
Endogenous detection of Phospho IRF7 in response to type I interferons
THP-1 cells were seeded in a 96-well plate (400,000 cells/well) in complete medium and incubated for approximately 10 minutes at 37°C, 5% CO2. The cells were treated with increasing concentrations of IFNα or IFNβ at the indicated concentrations for 6 hours.
After treatment, the cells were washed with HBSS and lysed with 50 µL of Lysis Buffer for 10 minutes at RT with shaking (350 rpm). IRF7 Phospho (Ser477) and Cofilin Total levels were evaluated using respective AlphaLISA SureFire Ultra assays. For the detection step, 10 µL of cell lysate (approximately 80,000 cells for Phospho IRF7 and 8,000 cells for Cofilin) was transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor mix and incubated for 1 hour at RT. Finally, 5 µL of Donor mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
As expected, treatment with type I interferons triggered a dose-dependent increase in the levels of Phospho IRF7 (Ser477) with no change to Cofilin levels.
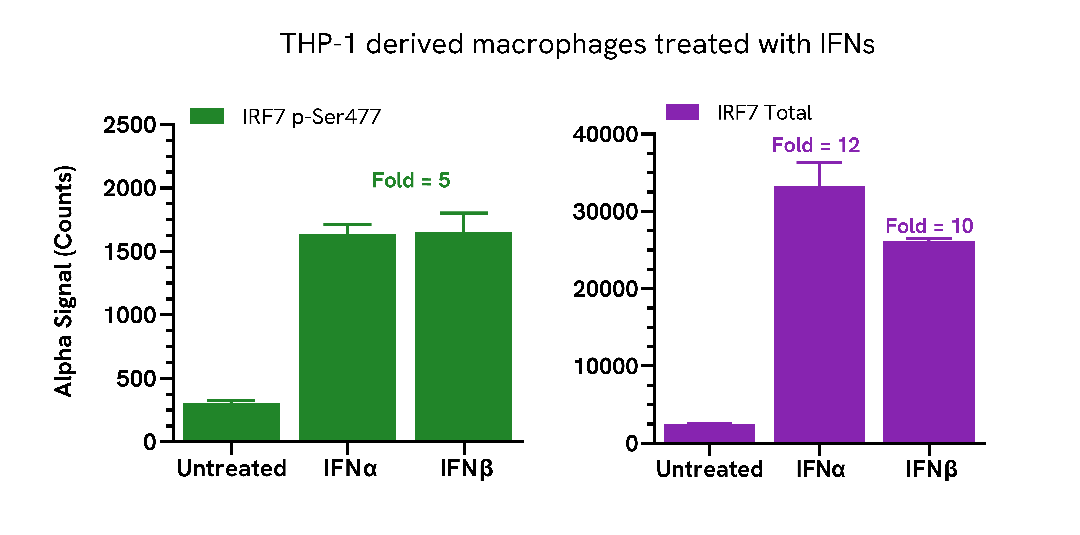
THP-1 cells were seeded in a 96-well plate (100,000 cells/well) in medium containing 100 nM PMA and incubated for 24 hours at 37°C, 5% CO2. THP-1 derived macrophages were then treated with 250 ng/mL of IFNα or IFNβ for a further 24 hours.
After treatment, the cells were washed with HBSS and lysed with 100 µL of Lysis Buffer for 10 minutes at RT with shaking (350 rpm). IRF7 Phospho (Ser477) and Total levels were evaluated using respective AlphaLISA SureFire Ultra assays. For the detection step, 10 µL of cell lysate (approximately 10,000 cells) was transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor mix and incubated for 1 hour at RT. Finally, 5 µL of Donor mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
As expected, treatment with type I interferons triggered an increase in the levels of Phospho (Ser477) and Total IRF7.
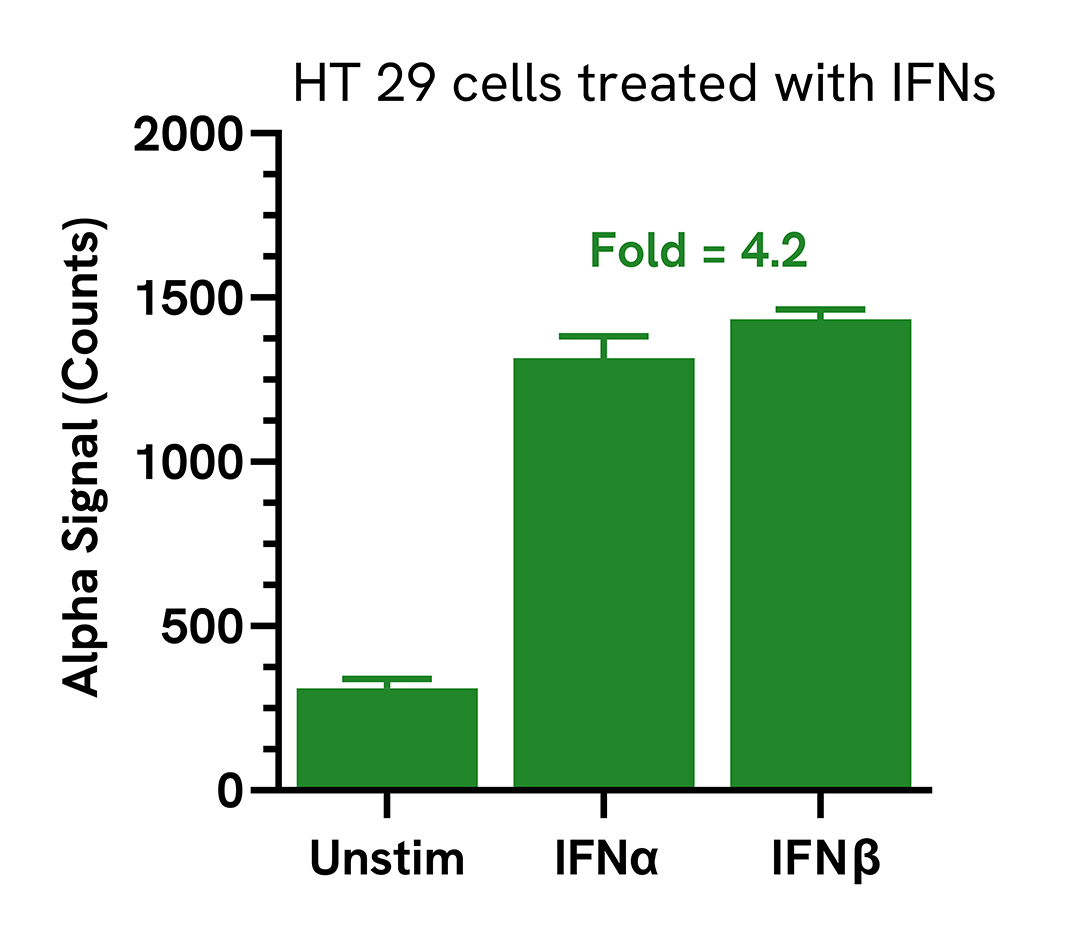
HT 29 cells were seeded in a 96-well plate (20,000 cells/well) in complete medium and incubated overnight at 37°C, 5% CO2. The cells were treated with 100 ng/mL of IFNα or IFNβ for 24 hours.
After treatment, the cells were washed with HBSS and lysed with 100 µL of Lysis Buffer for 10 minutes at RT with shaking (350 rpm). IRF7 Phospho (Ser477) levels were evaluated using the AlphaLISA SureFire Ultra assays. For the detection step, 10 µL of cell lysate (approximately 2,000 cells) was transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor mix and incubated for 1 hour at RT. Finally, 5 µL of Donor mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
Treatment with type I interferons triggered the activation of Phospho (Ser477) IRF7 in HT 29 cells.
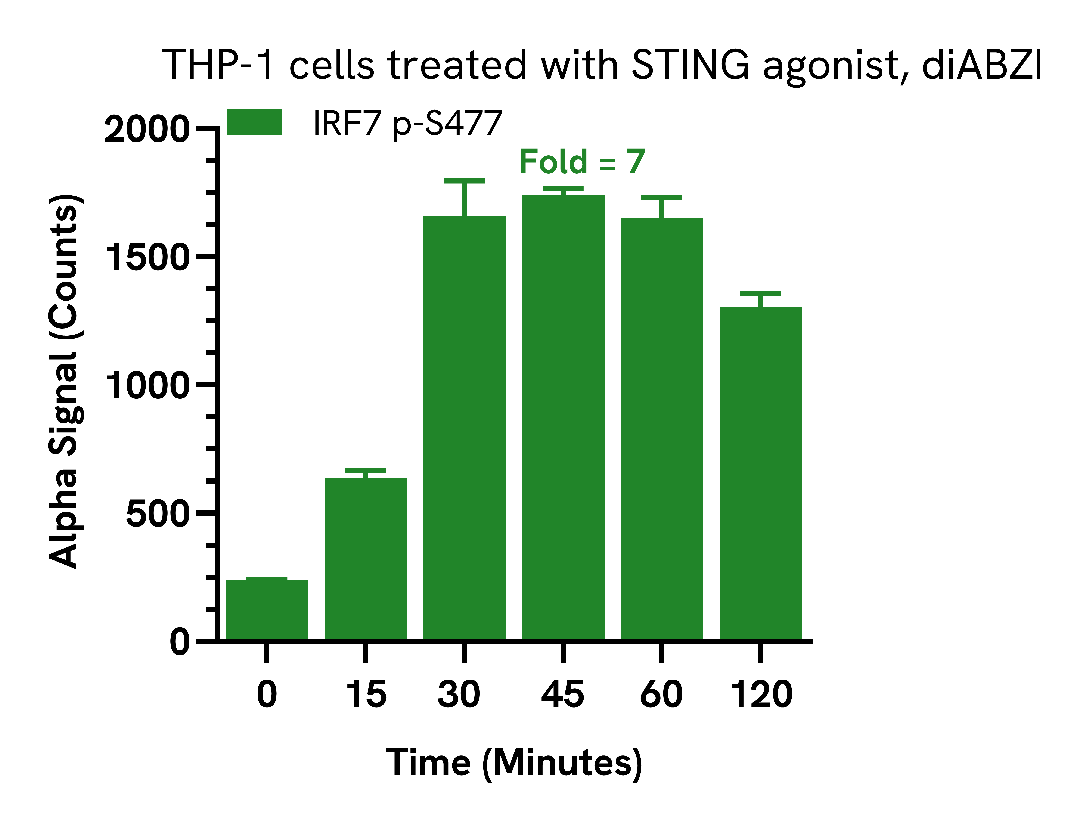
Activation of IRF7 by STING agonists
THP-1 cells were seeded in a 96-well plate (400,0000 cells/well) in complete medium and treated with 20 µM of STING agonist, diABZI at the indicated time points.
After treatment, the cells were washed with HBSS and then lysed with 100 µL of Lysis Buffer for 10 minutes at RT with shaking (350 rpm). IRF7 Phospho (Ser477) levels were evaluated using the AlphaLISA SureFire Ultra assays. For the detection step, 10 µL of cell lysate (approximately 40,000 cells) was transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor mix and incubated for 1 hour at RT. Finally, 5 µL of Donor mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
Treatment with STING agonist, diABZI, triggered a significant increase in IRF7 phosphorylation from 30 minutes with peak activation at 45 minutes.
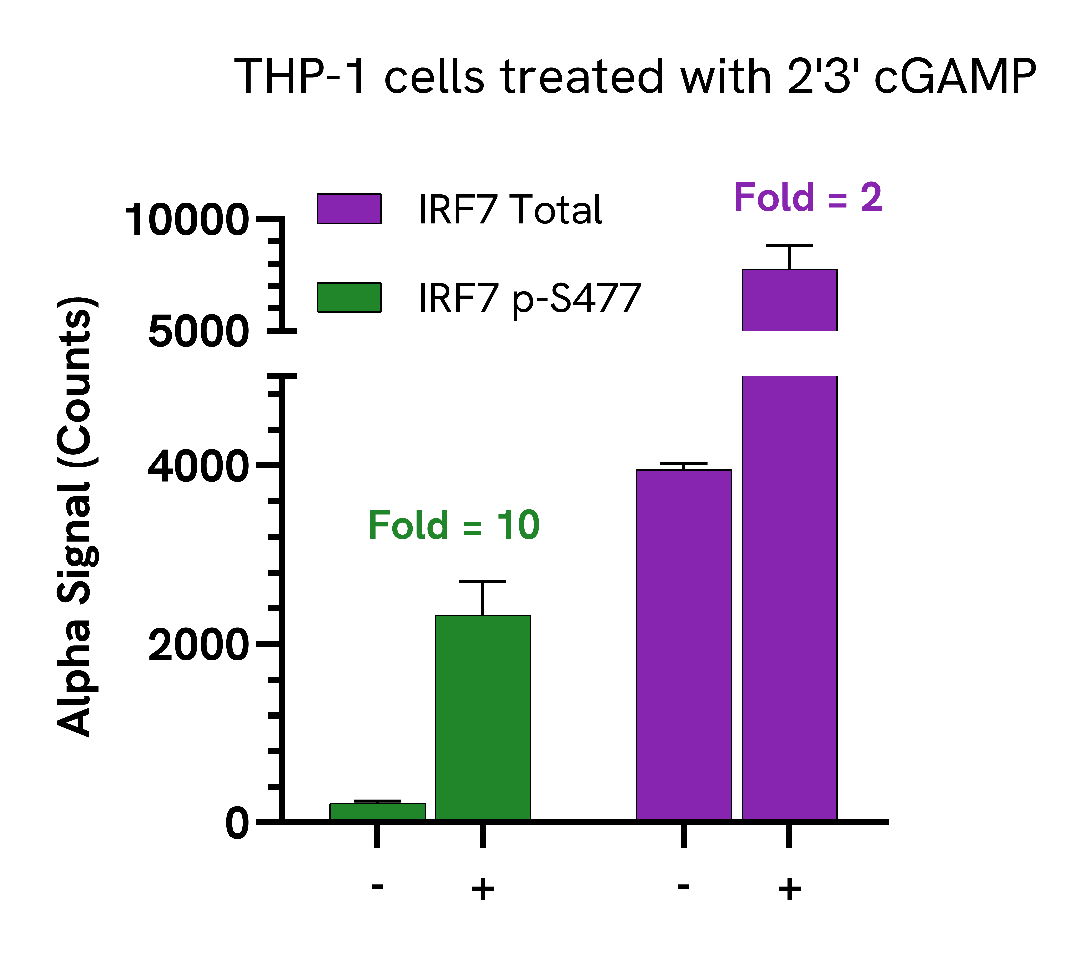
THP-1 cells were seeded in a 96-well plate (400,000 cells/well) in complete medium and treated with 100 µg/mL of STING ligand, 2’3’ cGAMP for 4 hours.
After treatment, the cells were washed with HBSS and then lysed with 100 µL of Lysis Buffer for 10 minutes at RT with shaking (350 rpm). IRF7 Phospho (Ser477) and Total levels were evaluated using the AlphaLISA SureFire Ultra assay. For the detection step, 10 µL of cell lysate (approximately 40,000 cells) was transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor mix and incubated for 1 hour at RT. Finally, 5 µL of Donor mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
Treatment with STING ligand, 2’3’ cGAMP, triggered a 10-fold increase of Phospho IRF7 (Ser477) and only 2-fold increase for Total IRF7.
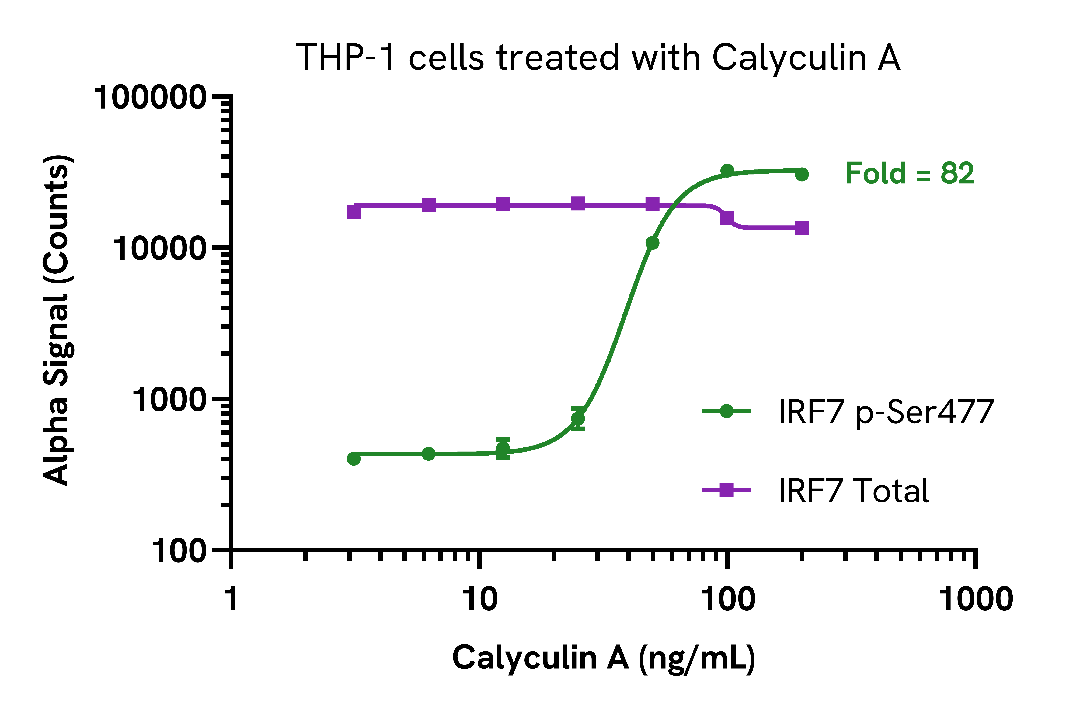
Activation of Phospho (Ser477) IRF7 in Calyculin A treated cells
THP-1 cells were seeded in a 96-well plate (100,000 cells/well) in medium containing 100 nM PMA and incubated for 18 hours at 37°C, 5% CO2. THP-1 derived macrophages were then treated with increasing concentrations of Calyculin A for 30 minutes.
After treatment, the cells were washed with HBSS and lysed with 100 µL of Lysis Buffer for 10 minutes at RT with shaking (350 rpm). IRF7 Phospho (Ser477) and Total levels were evaluated using respective AlphaLISA SureFire Ultra assays. For the detection step, 10 µL of cell lysate (approximately 10,000 cells) was transferred into a 384-well white OptiPlate, followed by 5 µL of Acceptor mix and incubated for 1 hour at RT. Finally, 5 µL of Donor mix was then added to each well and incubated for 1 hour at RT in the dark. The plate was read on an Envision using standard AlphaLISA settings.
As expected, treatment with Calyculin A triggered a dose-dependent increase in phosphorylated IRF7 (Ser477) with no change to IRF7 Total levels.
Specifications
| Application |
Cell Signaling
|
|---|---|
| Automation Compatible |
Yes
|
| Brand |
AlphaLISA SureFire Ultra
|
| Cellular or Signaling Pathway |
Inflammasome/Pattern Recognition Receptors (PRRs)
|
| Detection Modality |
Alpha
|
| Product Group |
Kit
|
| Protocol Time |
2h at RT
|
| Sample Volume |
10 µL
|
| Shipping Conditions |
Shipped in Blue Ice
|
| Target |
IRF7
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
|
| Technology |
Alpha
|
| Therapeutic Area |
Inflammation
Oncology
|
| Unit Size |
50,000 assay points
|
Resources
Are you looking for resources, click on the resource type to explore further.
Alpha technolgy enables the rapid and straightforward mesaure of virtually any target. This includes enzymes, receptor-ligand...
Discover Alpha SureFire® Ultra™ assays, the no-wash cellular kinase assays leveraging Revvity's exclusive bead-based technology...
This document includes detailed tables listing HTRF™, AlphaLISA™ SureFire® Ultra™, and Alpha SureFire® Ultra™ Multiplex assays...
Loading...


How can we help you?
We are here to answer your questions.






























